Effect of Pogostemon cablin Aqueous Extract on Gut Barrier in Mouse Model of Ulcerative Colitis
-
摘要: 目的:基于肠屏障研究广藿香水提物对溃疡性结肠炎(ulcerative colitis,UC)小鼠改善的作用及机制。方法:将小鼠随机分为空白组、模型组、美沙拉嗪组、广藿香水提物低、高剂量组;自由饮用2.5%葡聚糖硫酸钠(dextran sodium sulfate,DSS)10 d诱导小鼠UC模型,造模同时灌胃干预;检测血清白介素6(interleukin-6,IL-6)、白介素1β(IL-1β)和肿瘤坏死因子α(tumor necrosis factor α,TNF-α)水平;苏木素-伊红和高碘酸-席夫染色观察结肠病理变化;免疫组化法检测结肠紧密连接蛋白1(znula ocludens-1,ZO-1)、闭合连接蛋白1(occludin-1)和粘蛋白2(mucin 2,MUC2)蛋白表达;高通量测序技术分析结肠内容物肠道菌群变化。结果:DSS诱导的小鼠经广藿香水提物干预后,血清IL-6、IL-1β和TNF-α水平明显降低(P<0.05,P<0.01);结肠炎性浸润、上皮细胞结构破坏和杯状细胞数量减少等病理得到改善;结肠ZO-1、occludin-1和MUC2蛋白表达明显升高(P<0.05,P<0.01);肠道菌群α多样性sobs、Shannon和heip指数明显升高(P<0.05,P<0.01);肠道菌群β多样性主成分分析、主坐标分析和非度量多维尺度分析趋于健康小鼠;肠道菌群属水平优势物种隆布茨菌属(Romboutsia)、双歧杆菌属(Bifidobacterium)、布劳提菌属(Blautia)和unclassified_f__Lachnospiraceae丰度明显纠正(P<0.05,P<0.01)。结论:广藿香水提物对于DSS诱导的UC小鼠具有明确的改善作用,可能是其通过改善肠屏障而实现。Abstract: Objective: To explore the ameliorative effects and mechanisms of Pogostemon cablin aqueous extract (PCAE) on mice with ulcerative colitis (UC) based on gut barrier research. Methods: Mice were randomly divided into five groups: Control, model, mesalazine, low-dose of PCAE and high-dose of PCAE groups. A UC model was induced in mice by freely drinking 2.5% dextran sodium sulfate (DSS) for 10 d, with simultaneous oral administration of interventions. The study measured levels of serum interleukin-6 (IL-6), interleukin-1β (IL-1β), and tumor necrosis factor α (TNF-α). Pathological alterations in the colon were observed using hematoxylin-eosin and periodic acid-schiff staining. The expression of the colonic tight junction proteins zonula occludens-1 (ZO-1), occludin-1, and mucin 2 (MUC2) were assessed via immunohistochemistry. High-throughput sequencing technology was utilized to analyze changes in the gut microbiota of colonic contents. Results: Following intervention with PCAE, DSS-induced mice exhibited a significant reduction in serum levels of IL-6, IL-1β, and TNF-α (P<0.05, P<0.01). Pathological improvements in colon inflammation, epithelial cell structural damage, and reduced goblet cell numbers were observed. Additionally, the expression of colonic ZO-1, occludin-1, and MUC2 proteins significantly increased (P<0.05, P<0.01). Furthermore, the α-diversity indices of the gut microbiota including sobs, Shannon, and heip exhibited a marked increase (P<0.05, P<0.01), while the β-diversity analysis through principal component analysis, principal co-ordinates analysis, and non-metric multidimensional scaling analysis tended towards that of healthy mice. Notably, there was a significant correction in the abundance of dominant species at the genus level, including Romboutsia, Bifidobacterium, Blautia, and unclassified_f__Lachnospiraceae (P<0.05, P<0.01). Conclusion: PCAE exhibited a clear ameliorative effect on DSS-induced UC in mice, potentially achieved through the improvement of the gut barrier.
-
溃疡性结肠炎(ulcerative colitis,UC)是一种慢性非特异性炎症性肠病,其临床表现为反复性腹泻,便血,呕吐等症状[1−2]。现今,UC尚无根治的方法,临床上以抑制结肠粘膜的炎症为主要治疗思路,常用的治疗手段包括氨基水杨酸类、糖皮质激素、免疫抑制剂和生物制剂等药物治疗,以及外科手术治疗[3−4]。然而,这些治疗方法效果有限,并且可能伴有腹泻加重、骨质疏松、免疫抑制及术后感染等副作用[5]。因此,寻找新的UC干预手段仍十分迫切。保健食品具备特定功能特性,更为温和,因此有潜力成为UC干预策略的一部分。
肠屏障由紧密连接的肠上皮细胞、黏液层、免疫细胞和肠道菌群组成,这些结构协同维持肠道稳定[6]。UC发病机制尚不明确,但肠屏障损伤在UC的进展中起关键作用,具体表现为肠上皮结构破坏和肠道菌群紊乱引起的持续性炎症[6]。肠上皮结构破坏会导致肠道通透性增加,使得肠腔中的有害菌及其代谢产物能够渗透至肠粘膜下层,诱发局部免疫系统的激活以及炎症介质的释放[7]。正常生理状态下,肠道菌群能够发挥抗炎和维持免疫平衡的作用。然而,紊乱的肠道菌群表现出多样性降低和有害菌的过度生长,导致这些免疫调节机制失调并激活肠免疫细胞,进一步释放细胞因子和趋化因子,加剧组织损伤[8]。有研究表明,益生菌Lactobacillus plantarum HNU082能够通过修复肠屏障功能改善UC[9]。因此,修复肠屏障已成为改善UC的有效途径。
广藿香为唇形科植物广藿香Pogostemon cablin的干燥地上部分,具有芳香化浊,和中止呕,发表解暑之效力[10]。现代研究表明,广藿香具有抗炎、抗氧化、调节胃肠道和调节免疫等诸多药理及保健作用[11]。目前上市的国产保健食品如和中胶囊(国食健字G20041173)、三七砂仁软胶囊(国食健注G20130379)和维达软胶囊(国食健注G20160434)等均含有广藿香或其提取部位,表明了广藿香较好的食用保健价值。广藿香油和其部分有效成分展现出对UC的作用,据报道,广藿香油肠溶滴丸能够改善UC大鼠炎症水平[12]。此外,广藿香中的广藿香醇能够改善UC的炎症及肠上皮细胞的紧密连接[13]。广藿香中的β-广藿烯通过调节肠道菌群改善UC小鼠[14]。然而,广藿香水提物对于UC的作用及机制尚不清楚。因此,本研究采用葡聚糖硫酸钠(dextran sodium sulfate,DSS)诱导UC小鼠模型,探究广藿香水提物对其作用,并通过肠屏障研究机制,以期为广藿香在医疗和保健中用于改善UC提供依据。
1. 材料与方法
1.1 材料与仪器
雄性C57BL小鼠 6周龄50只,体重(21~25 g),江苏集萃药康生物科技股份有限公司提供(动物生产许可证号:SCXK(苏)2018-0008);广藿香饮片 安徽万生中药饮片公司;美沙拉嗪(原料药) 美国APExBIO公司;葡聚糖硫酸钠 美国MP公司;苏木素-伊红(hematoxylin-eosin,HE)和高碘酸-席夫(periodic acid-Schiff,PAS)染色试剂盒、白介素6(interleukin-6,IL-6)ELISA试剂盒、紧密连接蛋白1(znula ocludens-1,ZO-1)、粘蛋白2(mucin 2,MUC2) 武汉赛维尔生物公司;白介素1β(IL-1β)试剂盒、肿瘤坏死因子α(tumor necrosis factor α,TNF-α)ELISA试剂盒 泉州市睿信生物公司;闭合连接蛋白1(occludin-1) 英国Abcam公司;E.Z.N.A.® soil DNA、AxyPrep DNA凝胶提取、NEXTFLEX®快速DNA测序试剂盒 上海美吉生物公司。
万分之一电子天平 美国Mettler Toledo公司;Milli-Q实验室纯水系统 德国Merck公司;R-210型旋转蒸发仪 瑞士Buchi公司;SpectraMax iD3型多功能酶标仪 美国Molecular Devices公司;LEICA DM 2000荧光正置显微镜 德国Leica公司。
1.2 实验方法
1.2.1 广藿香水提物制备
广藿香以10倍水浸泡30 min,随后100 ℃煎煮30 min,过滤;滤渣以8倍水煎煮30 min,过滤;两次滤液合并,浓缩得到生药量为2.0 g/mL广藿香水提物母液,保存备用,使用前稀释。
1.2.2 UC小鼠模型构建及分组
本研究经江苏卫生健康职业学院实验动物伦理委员会批准(批准号:JHVC-IACUC-2022-B007)。小鼠适应性喂养7 d后,随机分为5组,每组10只:空白组、模型组、美沙拉嗪组、广藿香水提物低、高剂量组。除空白组小鼠外,其余各组2.5% DSS自由饮用10 d以诱导小鼠UC模型[6]。造模同时灌胃干预,美沙拉嗪组给予0.31 g/kg美沙拉嗪(按成人1.5 g/d用量换算,小鼠剂量为成人的12.3倍[15]),低剂量组和高剂量组分别给予0.62和2.05 g/kg上述广藿香水提物(广藿香成人用量范围为3~10 g/d[16],本研究初步以3 g对应剂量为低剂量,10 g对应剂量为高剂量,小鼠剂量为成人的12.3倍[15]),空白组和模型组以等量生理盐水干预。实验过程中,模型组小鼠死亡4只,低剂量组死亡2只,美沙拉嗪组死亡2只,高剂量组死亡1只,原因可能与2.5% DSS诱导的剧烈肠道炎症有关。实验终点时,模型组只剩6只小鼠,故其余各组均随机取6只小鼠进行实验,以保持每组小鼠数量一致。
1.2.3 样品采集
末次灌胃结束后,禁食不禁水12 h,眼眶采血,室温静置2 h,3000 r/min离心10 min,取上清,保存备用。用异氟醚麻醉后脱颈处死,打开腹腔取出结肠,快速收集结肠内容物于无菌冻存管中,并于液氮中速冻,用于后续肠道菌群测序;一部分新鲜结肠组织置于4%多聚甲醛中固定,用于后续病理检查;一部分新鲜结肠组织快速置于液氮中速冻,用于后续分子生物学检测。
1.2.4 小鼠炎性因子水平检测
血清中IL-6、IL-1β和TNF-α水平参照相应测试试剂盒说明书测定。
1.2.5 小鼠结肠组织病理形态学染色
小鼠结肠固定24 h后按标准操作流程脱水后包埋,随后切成5 μm的切片,浸入二甲苯中脱蜡及不同浓度乙醇进行水化,按照各染色说明书进行HE和PAS染色后置于光学显微镜下观察。
1.2.6 小鼠结肠组织ZO-1、occludin-1和MUC2免疫组织化学染色
取“1.2.5项”下组织切片脱蜡、水化后置于修复盒中,加入适量抗原修复液修复。冷却至室温后,洗涤3次,每次3 min,随后滴加100 μL过氧化氢溶液(3%),室温孵育15 min。孵育结束后,洗涤3次,每次3 min,加入适量山羊血清,封闭20 min。弃去血清,加入100 μL的1:200稀释的ZO-1、occludin-1和MUC2抗体,4 ℃过夜孵育。孵育结束后,回收一抗,洗涤3次,每次3 min,加入适量二抗,再加入苏木素复染3~5 min,随后使用超纯水冲洗2 min,封片后置于显微镜下观察,并采用ImageJ软件进行定量分析。
1.2.7 小鼠结肠内容物肠道菌群DNA抽提和PCR扩增
每组随机取5个样本进行测序。E.Z.N.A.® soil DNA提取试剂盒提取结肠内容物总DNA,1%琼脂糖凝胶电泳和紫外分光光度法评价提取质量、浓度和纯度,使用338F和806R引物对16S rRNA基因V3~V4可变区进行PCR扩增,扩增程序、反应体系与文献[17]报道一致。
1.2.8 小鼠结肠内容物肠道菌群Illumina Miseq测序
PCR产物混合后使用2%琼脂糖凝胶回收,并经过AxyPrep DNA凝胶提取试剂盒纯化,2%琼脂糖凝胶电泳检测及定量,使用NEXTFLEX®快速DNA测序试剂盒建库,导入上海美吉生物公司Miseq PE300平台进行测序,使用Trimmomatic和Flash对原始测序序列进行质控和拼接。测序数据于美吉生信云(https://cloud.majorbio.com/)处理,与文献[17]报道一致。
1.3 数据处理
数据处理使用SPSS 21.0,以平均值±标准差表示。单因素方差分析用于多组间比较分析,LSD-t(方差齐性)和Dunnett's T3(方差不齐)用于两组间比较分析,P<0.05表示具有显著性差异。
2. 结果与分析
2.1 广藿香水提物对UC小鼠炎性因子水平的影响
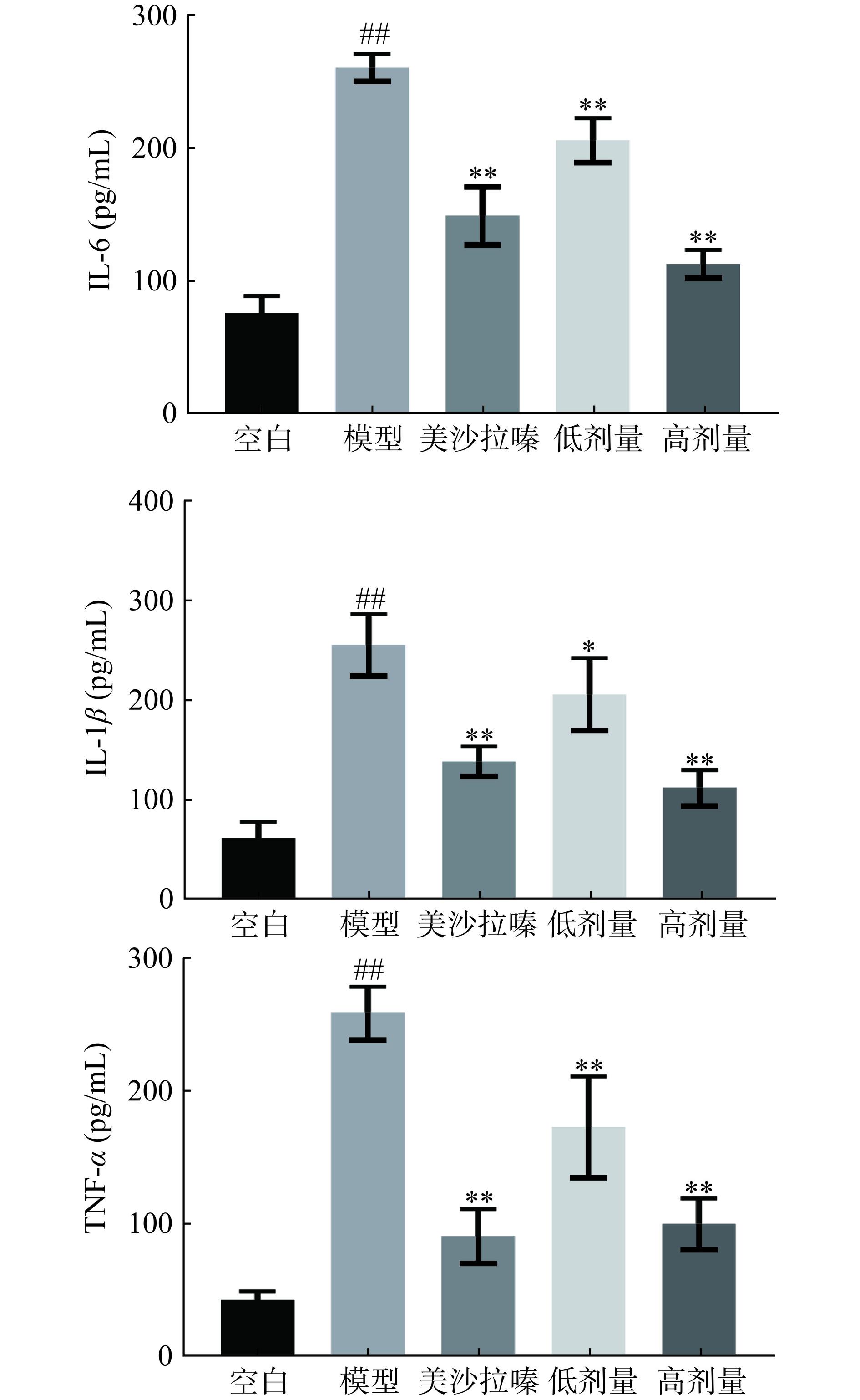
炎性因子包括IL-6、IL-1β和TNF-α等,它们在炎症过程中发挥重要作用,这些炎性因子水平可以反映机体炎症反应的强度,因此用试剂盒检测小鼠血清中炎性因子的水平。如图1所示,与空白组比较,模型组IL-6、IL-1β和TNF-α水平极显著升高(P<0.01);与模型组比较,美沙拉嗪、广藿香水提物低剂量和高剂量组IL-6、IL-1β和TNF-α水平显著降低(P<0.05,P<0.01),且高剂量广藿香效果与美沙拉嗪相当。这些结果与马广礼等[18]和董晶等[19]研究一致,说明广藿香水提物具有改善UC小鼠炎症的作用。
2.2 广藿香水提物对UC小鼠结肠病理组织损伤的影响
肠道病理检查如HE染色和PAS染色,可以全面评估UC小鼠模型的肠道病理变化,为疾病的研究提供重要的信息。如图2所示,空白组小鼠结肠上皮结构完整;模型组小鼠结肠出现炎性浸润,上皮结构破坏严重,杯状细胞数量减少;广藿香水提物低剂量组小鼠结肠上皮结构及杯状细胞数量稍有恢复,炎性浸润稍有改善;广藿香水提物高剂量组小鼠结肠上皮结构趋于正常,杯状细胞数量大量恢复,炎性浸润不明显。高剂量广藿香改善效果与美沙拉嗪接近。这些结果与马广礼等[18]和董晶等[19]研究一致,说明广藿香水提物具有改善UC小鼠结肠病理的作用。
2.3 广藿香水提物对UC小鼠结肠上皮的影响
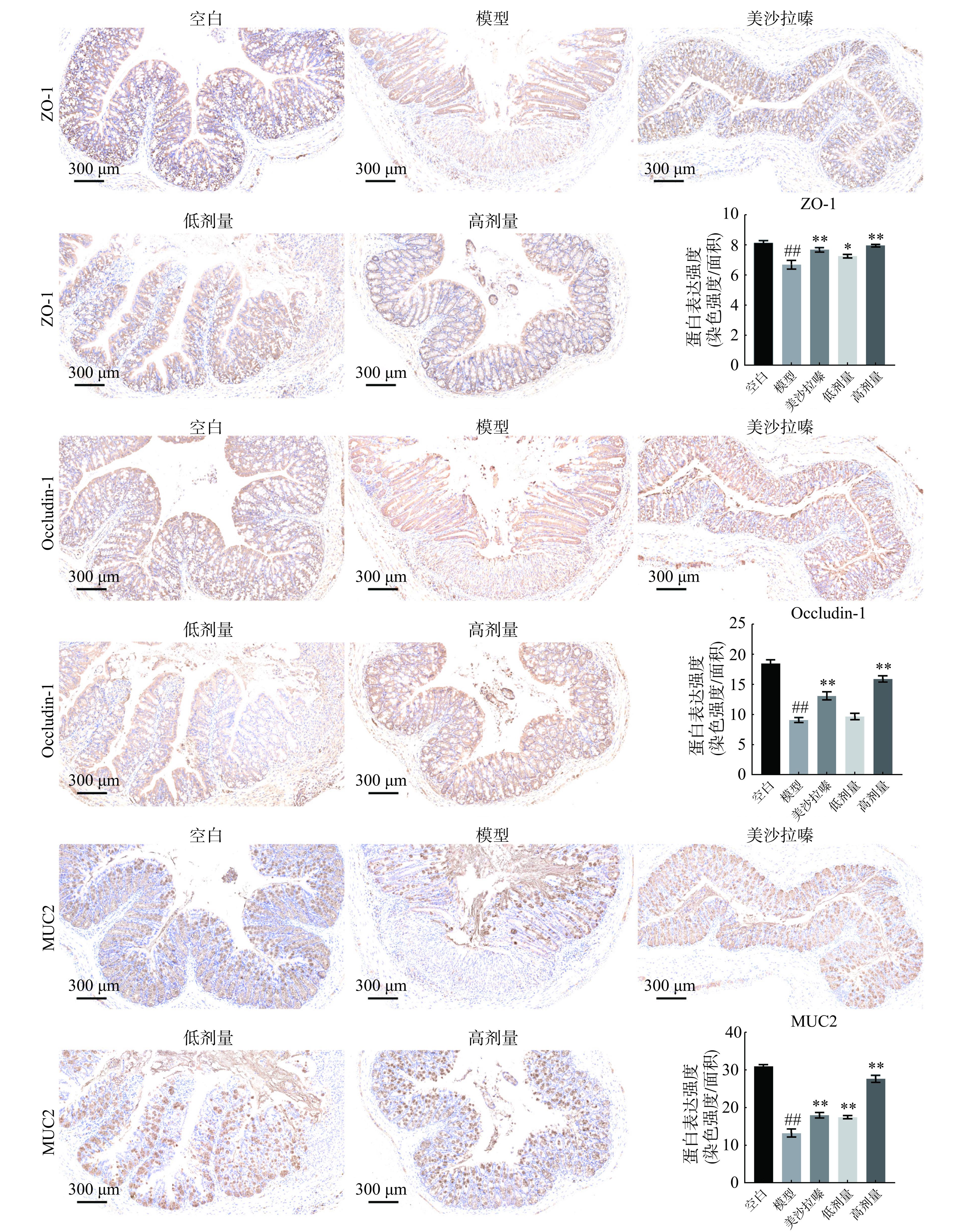
紧密连接蛋白包括ZO-1和occludin-1是肠上皮结构稳定的基础,能够反映肠道健康情况。肠道粘液有助于维持肠道正常的生理活动,其中主要成分为MUC2。如图3所示,与空白组比较,模型组小鼠结肠ZO-1、occludin-1和MUC2蛋白表达极显著下降(P<0.01);与模型组比较,广藿香水提物高剂量组小鼠结肠ZO-1、occludin-1和MUC2蛋白表达极显著升高(P<0.01),升高效果优于美沙拉嗪;与模型组比较,广藿香水提物低剂量组小鼠结肠ZO-1和MUC2蛋白表达显著升高(P<0.05,P<0.01)。这些结果与Yan等[6]研究一致,说明广藿香水提物具有改善UC小鼠肠上皮屏障的作用。
2.4 广藿香水提物对UC小鼠肠道菌群α多样性的影响
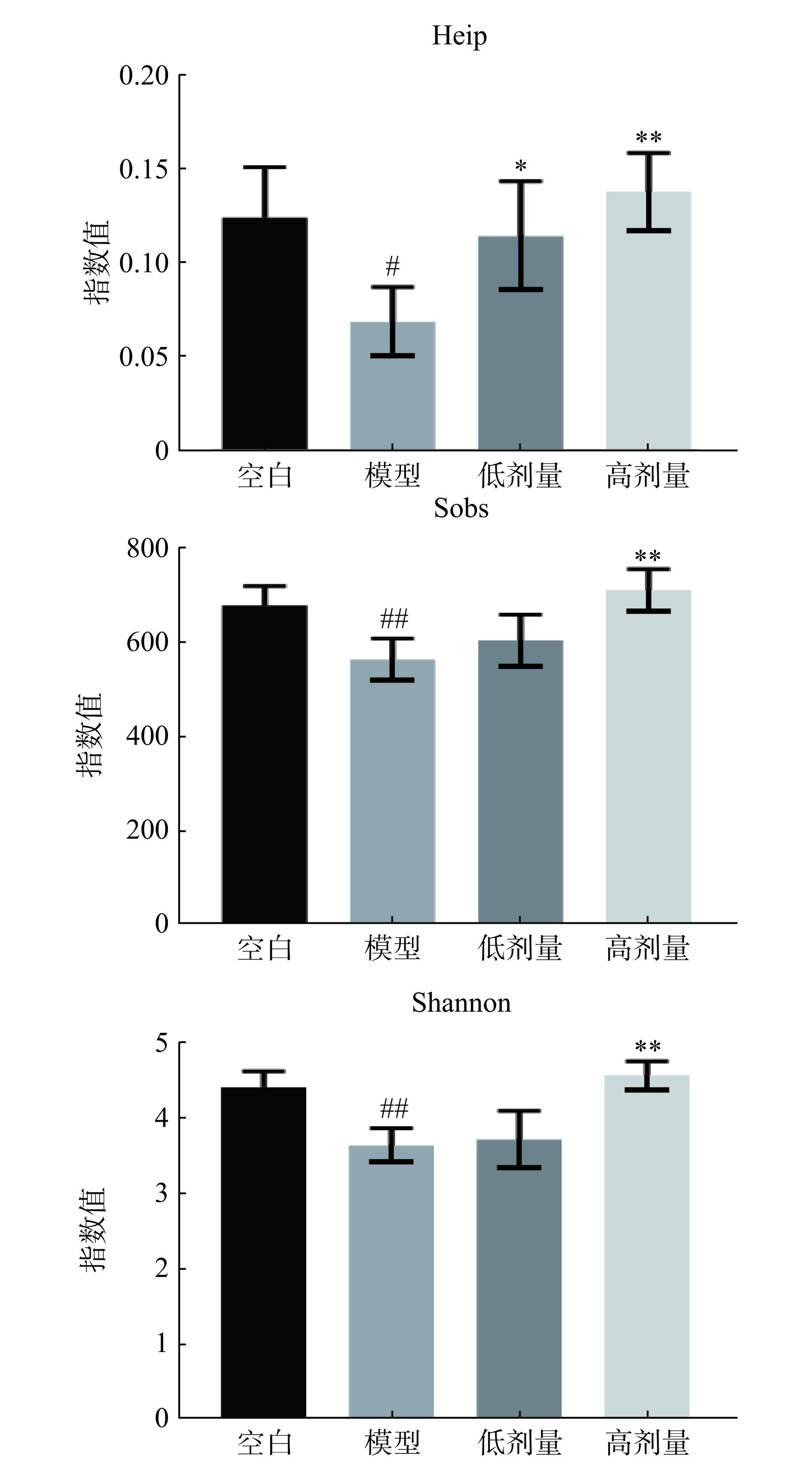
肠道菌群α多样性分析对于了解肠道微生物群落的生态系统和稳态具有重要意义,其中sobs(代表样本中观察到的菌群数量)、shannon(与物种丰富度和均匀度正相关)和heip(与物种不平衡和失调相关)指数能够反映微生物的丰富度和均匀度,进而显示UC患者及实验动物模型肠道菌群的多样性水平和群落的稳定性。如图4所示,与空白组比较,模型组α多样性sobs、shannon和heip指数显著下降(P<0.05,P<0.01);与模型组比较,广藿香水提物高剂量组三种指数极显著升高(P<0.01),广藿香水提物低剂量组heip指数显著升高(P<0.05),其余指数具有一定升高趋势,但无统计学意义(P>0.05)。这些结果与文献[6,20]研究一致,说明广藿香水提物高剂量组具有改善UC小鼠肠道菌群α多样性的作用。
2.5 广藿香水提物对UC小鼠肠道菌群β多样性的影响
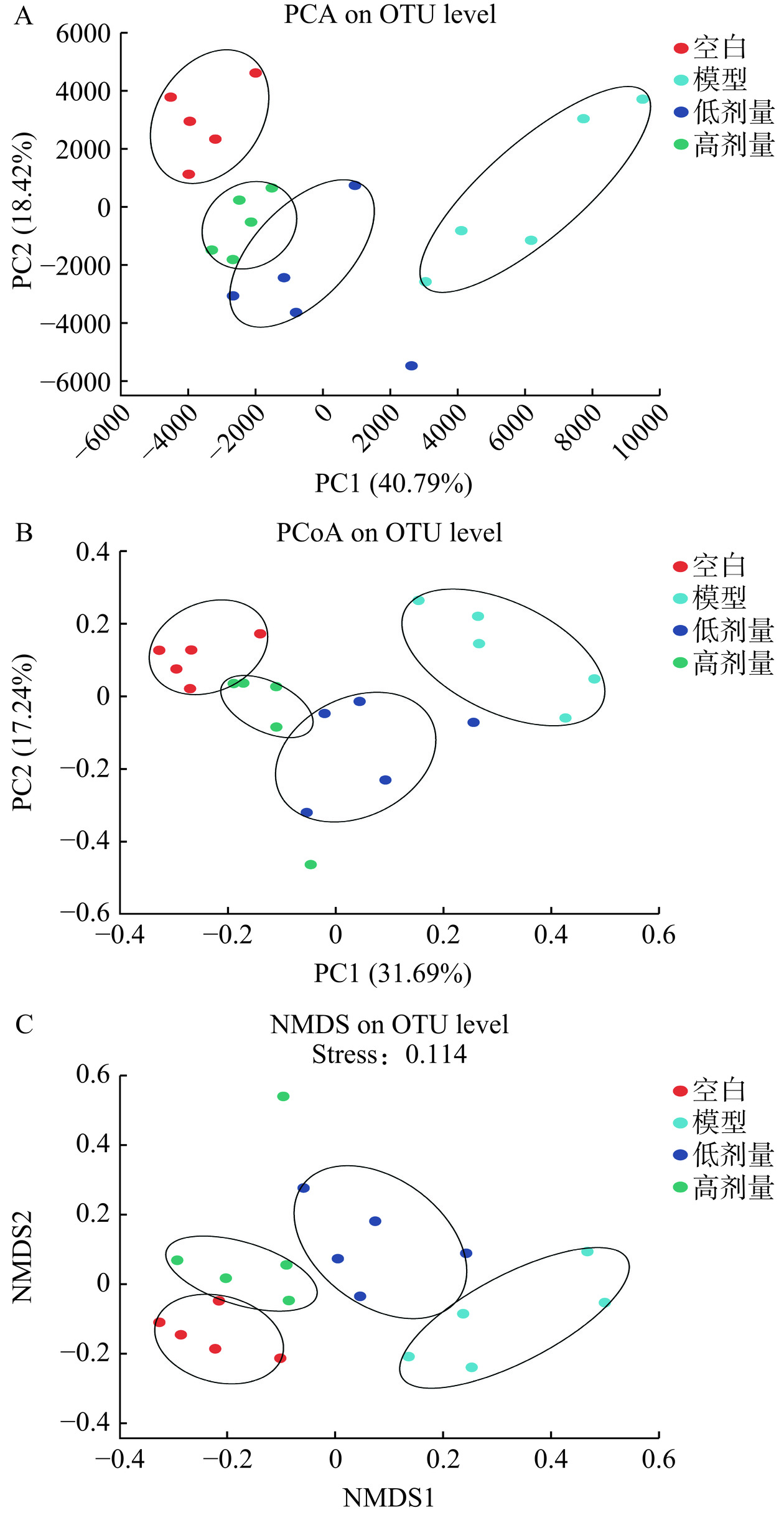
肠道菌群β多样性分析对于了解肠道微生物群落之间的相似性和差异性至关重要,主成分分析(principal component analysis,PCA)、主坐标分析(principal co-ordinates analysis,PCoA)和非度量多维尺度分析(non-metric multidimensional scaling analysis,NMDS)为其常用分析方法。如图5所示,在肠道菌群β多样性中的PCA、PCoA和NMDS中,模型组与其余三组聚集圈距离较远,广藿香水提物高剂量组与空白组聚集圈距离接近,说明模型组与其余三组肠道菌群β多样性差异较大,空白组与广藿香高剂量组肠道菌群β多样性较为相似。这些结果与文献[6,20]研究一致,说明广藿香水提物具有改善UC小鼠肠道菌群β多样性的作用。
2.6 广藿香水提物对UC小鼠肠道菌群物种组成的影响
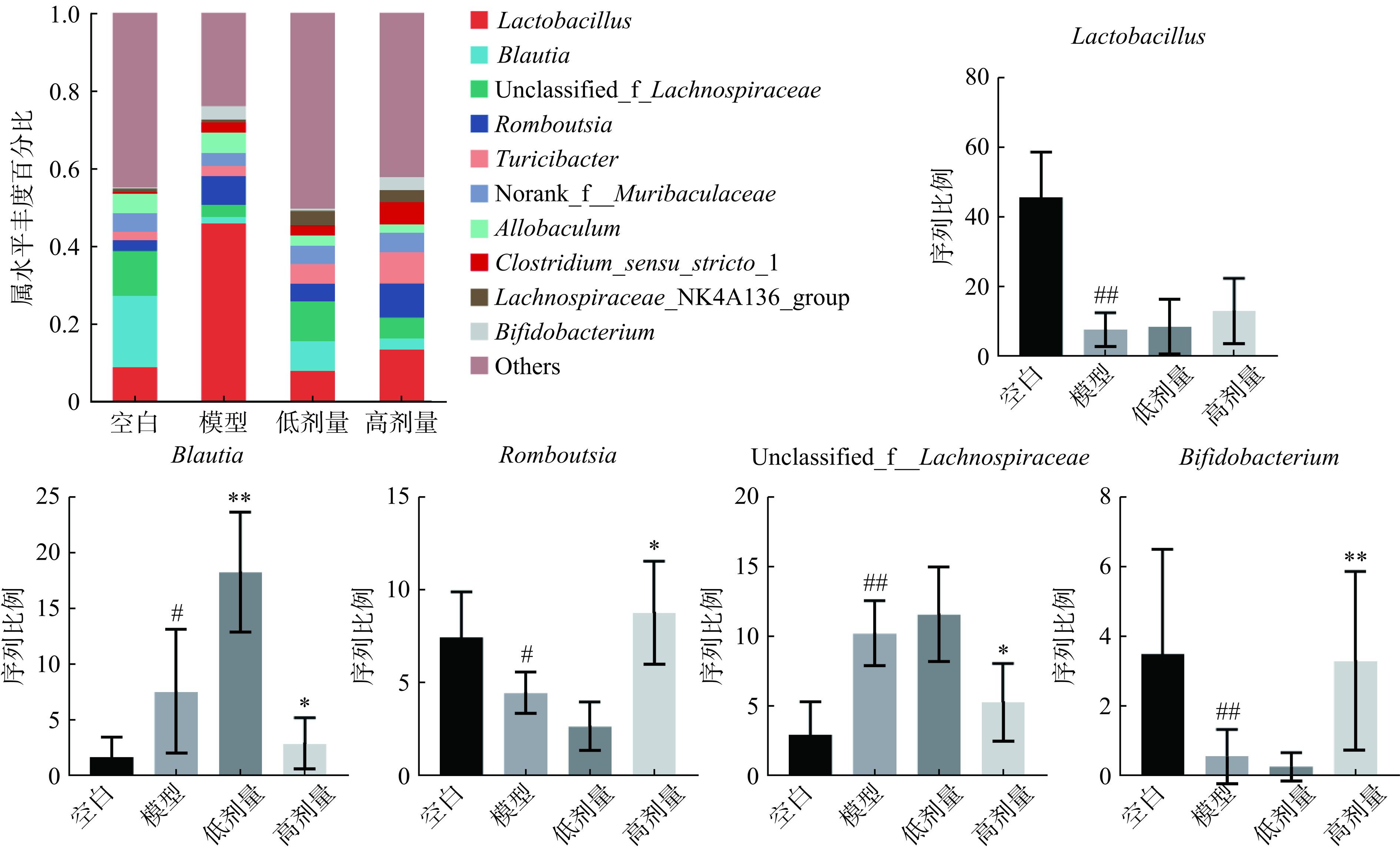
肠道菌群属水平物种丰度的变化可能在炎症调节、黏膜保护和免疫调节等多个方面影响UC的病理生理过程。如图6所示,各组小鼠在属水平群落丰度排名前十的优势物种分别为乳酸杆菌属(Lactobacillus)、布劳提菌属(Blautia)、unclassified_f_Lachnospiraceae、隆布茨菌属(Romboutsia)、土里氏杆菌属(Turicibacter)、norank_f_Muribaculaceae、异球菌属(Allobaculum)、Clostridium_sensu_stricto_1、Lachnospiraceae_NK4A136_group和双歧杆菌属(Bifidobacterium)。在这些优势物种中,与空白组比较,模型组乳酸杆菌属、隆布茨菌属和双歧杆菌属丰度显著下降(P<0.05,P<0.01),布劳提菌属和unclassified_f_Lachnospiraceae丰度显著升高(P<0.05,P<0.01);与模型组比较,广藿香水提物高剂量组隆布茨菌属和双歧杆菌属丰度显著上升(P<0.05,P<0.01),乳酸杆菌属丰度有上升趋势,布劳提菌属和unclassified_f__Lachnospiraceae丰度显著下降(P<0.05);与模型组比较,低剂量组乳酸杆菌属丰度有上升趋势,但差异无统计学意义(P>0.05)。据报道,乳酸杆菌属、隆布茨菌属和双歧杆菌属与UC进展有关,其丰度增加与疾病严重程度呈负相关[21−25]。与这些研究一致,广藿香水提物能够通过纠正乳酸杆菌属、隆布茨菌属和双歧杆菌属等属水平物种丰度改善UC小鼠。
2.7 肠道菌群属水平优势物种与炎症水平及肠上皮结构的相关性分析
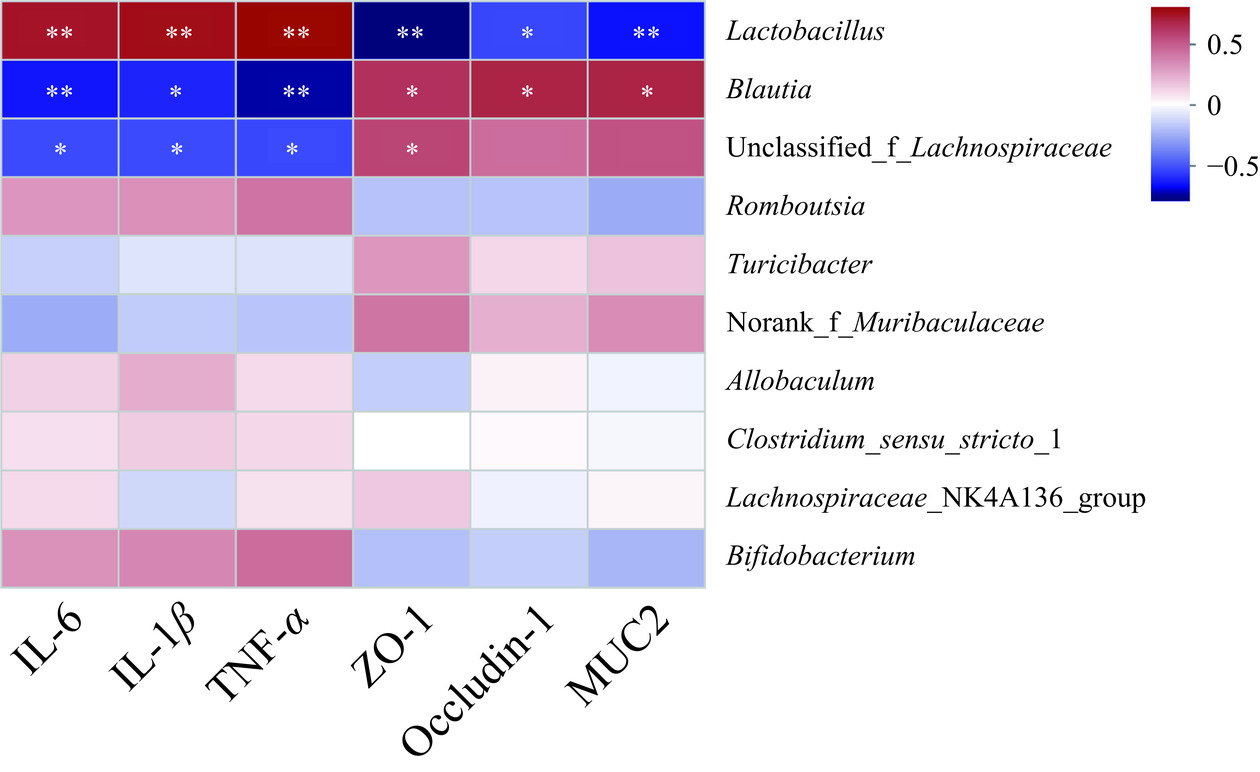
相关性分析能够发现特定菌群与干预效果之间的潜在关系,这能够进一步揭示干预措施的科学内涵。如图7所示,乳酸杆菌属与IL-6、IL-1β和TNF-α呈极显著正相关(P<0.01),与ZO-1、occludin-1和MUC2呈显著负相关(P<0.05,P<0.01);布劳提菌属与IL-6、IL-1β和TNF-α呈显著负相关(P<0.05,P<0.01),与ZO-1、occludin-1和MUC2呈正相关(P<0.05);unclassified_f_Lachnospiraceae与IL-6、IL-1β和TNF-α呈显著负相关(P<0.05),与ZO-1呈正相关(P<0.05)。这些结果与Yan等[6]研究一致,说明肠道菌群属水平物种与UC的炎症及肠上皮结构有关,提示广藿香水提物能够通过调控乳酸杆菌属、布劳提菌属和unclassified_f_Lachnospiraceae等属水平物种丰度调控UC小鼠的炎症和肠上皮结构。
3. 讨论与结论
DSS诱导小鼠UC是一种常用的动物模型,通过给小鼠饮用不同浓度的DSS,引起结肠黏膜的炎症、溃疡和屏障功能损害,这些病理与UC患者的临床症状高度相似[26]。所以,这种模型广泛用于UC的发病机制、治疗策略以及疗效的评估[26]。美沙拉嗪具有明确的抗炎作用,是UC临床用药,所以作为本研究的阳性对照。本研究中,DSS诱导的小鼠血清炎性因子水平明显上升,结肠黏膜出现炎性浸润,结肠上皮结构破坏严重,杯状细胞数量减少,这些结果表明DSS诱导小鼠UC模型构建成功。含广藿香成分的功能食品作为一种非药物干预手段,具有调节人体生理功能、改善健康状况和预防疾病的功能[11]。在研究中,给予UC小鼠不同浓度广藿香水提物干预后,高剂量组小鼠血清炎性因子水平明显减低,结肠粘膜炎性浸润改善,结肠上皮及杯状细胞结构恢复,低剂量组的小鼠血清炎性因子水平减低也具有统计学意义,结肠粘膜炎性浸润、上皮及杯状细胞结构也出现一定改善趋势。这些结果均表明,广藿香水提物具有明确改善UC小鼠的作用。
肠屏障是指整个肠道防御系统,包括肠上皮屏障、肠道菌群和肠免疫屏障,在UC进展中扮演重要角色[6]。这其中,肠上皮屏障是由肠道上皮细胞形成的屏障,它覆盖在肠道黏膜表面,并通过紧密连接蛋白(ZO-1和occludin-1)和黏附分子(MUC2)等结构物质连接在一起[27]。UC患者肠上皮存在结构性损伤,肠上皮屏障受损,肠道通透性增加,导致一些有害物质(如细菌、毒素等)和大分子物质(如蛋白质)穿过肠道壁进入体内,随后加剧疾病[28]。研究证实,肠上皮细胞紧密连接蛋白等被破坏是以上改变的主要原因[29]。在本研究中,UC小鼠结肠组织ZO-1、occludin-1和MUC2蛋白表达明显降低,结合结肠病理结果表明,UC小鼠的肠上皮屏障受损。广藿香水提物干预后,小鼠结肠组织ZO-1、occludin-1和MUC2蛋白表达明显升高,综合病理结果表明,广藿香水提物能够改善UC小鼠的肠上皮屏障。
肠道微生物群和宿主之间存在着共生关系,能通过多种途径参与宿主代谢,直接或间接影响宿主的生理病理过程。肠道菌群失调会诱导身体的免疫反应,损伤肠道黏膜屏障能力,进而导致肠道炎症[30]。UC患者的肠道菌群与健康人相比存在明显的变化,调节肠道菌群已成为改善UC的重要手段[31]。功能食品如鱼腥草和抗性糊精等能够通过调节肠道菌群改善UC[32−33]。在本研究中,UC小鼠较健康小鼠肠道菌群的α和β多样性明显改变,而广藿香水提物干预后,小鼠肠道菌群多样性得到纠正,并趋于健康小鼠,这表明广藿香水提物具有调节UC小鼠肠道菌群的作用。此外,广藿香水提物干预后,在属水平优势物种上较UC小鼠肠道菌群乳酸杆菌属、布劳提菌属、unclassified_f__Lachnospiraceae、隆布茨菌属和双歧杆菌属趋于健康小鼠。乳酸杆菌属和双歧杆菌属是两类常见的益生菌,被广泛应用于调节肠道菌群平衡和维护肠道健康[21]。UC患者口服乳酸杆菌属和双歧杆菌属菌株能够一定程度改善临床症状和疾病活动度[22]。动物实验证实,乳酸杆菌属和双歧杆菌属可以抑制炎症反应,通过减少炎性细胞浸润、抑制炎症细胞因子的产生和调节免疫平衡,从而减轻UC引起的肠道炎症[23]。此外,乳酸杆菌属和双歧杆菌属有助于加强肠道屏障功能,通过增加粘膜黏液层和增强黏膜的完整性,减少病原菌和有害物质的侵袭[23−24]。隆布茨菌属与UC之间可能存在一定的关系,研究证实其可能通过调节短链脂肪酸的代谢来调节肠道炎症的发生和发展[25]。有研究报道广藿香水提物中含原儿茶酸、绿原酸、香草酸、咖啡酸等多种水溶性成分[34],其中原儿茶酸能够增加高脂膳食小鼠结肠中菌群丰度和多样性[35],抑制有害菌弯曲杆菌门的相对丰度,绿原酸能够调节DSS诱导的结肠炎小鼠肠道放线菌、乳酸菌、双歧杆菌、拟杆菌、亚芽胞杆菌和链球菌的丰度[36],咖啡酸降低了DSS诱导的结肠炎小鼠肠道拟杆菌和Turicibacter的相对丰度,提高了Alistipes、Dubosiella及阿克曼菌属的相对丰度[37]。因此,广藿香水提物能够发挥改善UC的作用可能是通过其中多种水溶性有效成分调节肠道菌群进而改善肠屏障而实现的。
本研究中,乳酸杆菌属与IL-6、IL-1β和TNF-α呈正相关,与ZO-1、occludin-1和MUC2呈负相关;布劳提菌属与IL-6、IL-1β和TNF-α呈负相关,与ZO-1、occludin-1和MUC2呈正相关;unclassified_f_Lachnospiraceae与IL-6、IL-1β和TNF-α呈负相关,与ZO-1呈正相关。说明肠道菌群属水平物种与UC的炎症及肠上皮结构有关,提示广藿香水提物能够通过调控乳酸杆菌属、布劳提菌属和unclassified_f_Lachnospiraceae等属水平物种丰度改善UC小鼠的炎症和肠上皮结构。
综上,广藿香水提物对于DSS诱导的UC小鼠具有明确的改善作用,这一作用可能是其通过提高UC小鼠肠道乳酸杆菌属及双歧杆菌属丰度,抑制炎症反应,增加肠道粘膜屏障完整性,同时通过增加隆布茨菌属丰度调节短链脂肪酸代谢进而调节肠道炎症。研究结果可为广藿香及其功能食品用于改善UC提供依据。
-
-
[1] ZUO T, LU X J, ZHANG Y, et al. Gut mucosal virome alterations in ulcerative colitis[J]. Gut,2019,68(7):1169−1179. doi: 10.1136/gutjnl-2018-318131
[2] WEI S C, SOLLANO J, HUI Y T, et al. Epidemiology, burden of disease, and unmet needs in the treatment of ulcerative colitis in Asia[J]. Expert Review of Gastroenterology & Hepatology,2021,15(3):275−289.
[3] 张帅, 刘英媛. 中医治疗溃疡性结肠炎的研究进展[J]. 中国实用乡村医生杂志,2022,29(5):29−32. [ZHANG S, LIU Y Y. Advances in the treatment of ulcerative colitis with traditional Chinese medicine[J]. Chinese Practical Journal of Rural Doctor,2022,29(5):29−32.] ZHANG S, LIU Y Y. Advances in the treatment of ulcerative colitis with traditional Chinese medicine[J]. Chinese Practical Journal of Rural Doctor, 2022, 29(5): 29−32.
[4] 陆伟, 陈红斗, 王梦雷, 等. 1例重度溃疡性结肠炎糖皮质激素抵抗患者的环孢素转换治疗[J]. 中国现代应用药学,2021,38(2):232−236. [LU W, CHEN H D, WANG M L, et al. Treatment of a case of severe ulcerative colitis with glucocorticoid resistance patient by cyclosporine conversion[J]. Chinese Journal of Modern Applied Pharmacy,2021,38(2):232−236.] LU W, CHEN H D, WANG M L, et al. Treatment of a case of severe ulcerative colitis with glucocorticoid resistance patient by cyclosporine conversion[J]. Chinese Journal of Modern Applied Pharmacy, 2021, 38(2): 232−236.
[5] 严宝飞, 许晨新, 陈灵, 等. 基于TLR2/MyD88/NF-κB信号通路探讨黄芩汤对溃疡性结肠炎小鼠的治疗机制[J]. 中药新药与临床药理,2022,33(6):727−735. [YAN B F, XU C X, CHEN L, et al. Study on the therapeutic mechanism of Huangqin Decoction on mice with ulcerative colitis through TLR2/MyD88/NF-κB signaling pathway[J]. Traditional Chinese Drug Research & Clinical Pharmacology,2022,33(6):727−735.] YAN B F, XU C X, CHEN L, et al. Study on the therapeutic mechanism of Huangqin Decoction on mice with ulcerative colitis through TLR2/MyD88/NF-κB signaling pathway[J]. Traditional Chinese Drug Research & Clinical Pharmacology, 2022, 33(6): 727−735.
[6] YAN B F, CHEN X, CHEN Y F, et al. Aqueous extract of Paeoniae Radix Alba (Paeonia lactiflora Pall.) ameliorates DSS-induced colitis in mice by tunning the intestinal physical barrier, immune responses, and microbiota[J]. Journal of Ethnopharmacology,2022,294:115365. doi: 10.1016/j.jep.2022.115365
[7] MEHANDRU S, COLOMBEL J F. The intestinal barrier, an arbitrator turned provocateur in IBD[J]. Nature Reviews Gastroenterology and Hepatology,2021,18(2):83−84. doi: 10.1038/s41575-020-00399-w
[8] ELSON CO, CONG Y. Host-microbiota interactions in inflammatory bowel disease[J]. Gut Microbes,2012,3(4):332−44. doi: 10.4161/gmic.20228
[9] WU Y, JHA R, LI A, et al. Probiotics (Lactobacillus plantarum HNU082) supplementation relieves ulcerative colitis by affecting intestinal barrier functions, immunity-related gene expression, gut microbiota, and metabolic pathways in mice[J]. Microbiology Spectrum,2022,10(6):e01651−22.
[10] 荆文光, 郭晓晗, 李楚, 等. 基于质量源于生产的广藿香质量标志物的确立[J]. 中草药,2021,52(15):4496−4506. [JING W G, GUO X H, LI C, et al. Establishment of quality markers for Pogostemonis Herba based on quality by production[J]. Chinese Traditional and Herbal Drugs,2021,52(15):4496−4506.] JING W G, GUO X H, LI C, et al. Establishment of quality markers for Pogostemonis Herba based on quality by production[J]. Chinese Traditional and Herbal Drugs, 2021, 52(15): 4496−4506.
[11] 李楚, 荆文光, 莫小路, 等. 广藿香化学成分和药理作用研究进展及潜在质量标志物预测分析[J]. 中国药学杂志,2023,58(11):954−965. [LI C, JING W G, MO X L, et al. Research progress in chemical constituents and pharmacological effects of pogostemon cablin and predictive analysis of quality marker[J]. Chinese Pharmaceutical Journal,2023,58(11):954−965.] LI C, JING W G, MO X L, et al. Research progress in chemical constituents and pharmacological effects of pogostemon cablin and predictive analysis of quality marker[J]. Chinese Pharmaceutical Journal, 2023, 58(11): 954−965.
[12] 李晓锋, 陈卫东, 陈华源, 等. 广藿香油肠溶滴丸制备及其对溃疡性结肠炎大鼠的药效评价[J]. 中国现代应用药学,2024,41(12):1621−1630. [LI X F, CHEN W D, CHEN H Y, et al. Preparation of patchouli oil enteric-coated dropping pills and its efficacy evaluation on ulcerative colitis rats[J]. Chinese Journal of Modern Applied Pharmacy,2024,41(12):1621−1630.] LI X F, CHEN W D, CHEN H Y, et al. Preparation of patchouli oil enteric-coated dropping pills and its efficacy evaluation on ulcerative colitis rats[J]. Chinese Journal of Modern Applied Pharmacy, 2024, 41(12): 1621−1630.
[13] WU Z N, ZENG H R, ZHANG L L, et al. Patchouli alcohol:A natural sesquiterpene against both inflammation and intestinal barrier damage of ulcerative colitis[J]. Inflammation,2020,43(4):1423−1435. doi: 10.1007/s10753-020-01219-8
[14] LIU Y H, WU J Z, CHEN L P, et al. β-patchoulene simultaneously ameliorated dextran sulfate sodium-induced colitis and secondary liver injury in mice via suppressing colonic leakage and flora imbalance[J]. Biochemical Pharmacology,2020,182:114260. doi: 10.1016/j.bcp.2020.114260
[15] 陈奇, 张伯礼. 中药药效研究方法学[M]. 北京:人民卫生出版社, 2016:10. [CHEN Q, ZHANG B L. Methodology of traditional Chinese medicine efficacy research[M]. Beijing:People’s Medical Publishing House, 2016:10.] CHEN Q, ZHANG B L. Methodology of traditional Chinese medicine efficacy research[M]. Beijing: People’s Medical Publishing House, 2016: 10.
[16] 国家药典委员会. 中华人民共和国药典(一部)[M]. 北京:中国医药科技出版社, 2020:46. [National Pharmacopoeia Committee. Pharmacopoeia of the People’s Republic of China (Volume One)[M]. Beijing:China Medical Science and Technology Press, 2020:46.] National Pharmacopoeia Committee. Pharmacopoeia of the People’s Republic of China (Volume One)[M]. Beijing: China Medical Science and Technology Press, 2020: 46.
[17] 严宝飞, 戴仕林, 刘圣金, 等. 黄芩汤对非酒精性脂肪肝炎大鼠的作用及机制研究[J]. 中国医院药学杂志,2022,42(10):1009−1014. [YAN B F, DAI S L, LIU S J, et al. Study on the effect and mechanism of Huangqin Decoction in non-alcoholic steatohepatitis rats[J]. Chinese Journal of Hospital Pharmacy,2022,42(10):1009−1014.] YAN B F, DAI S L, LIU S J, et al. Study on the effect and mechanism of Huangqin Decoction in non-alcoholic steatohepatitis rats[J]. Chinese Journal of Hospital Pharmacy, 2022, 42(10): 1009−1014.
[18] 马广礼, 夏晓培, 马金亮. 蒿本内酯对葡聚糖硫酸钠诱导的溃疡性结肠炎小鼠的改善作用[J]. 食品工业科技,2024,45(4):321−327. [MA G L, XIA X P, MA J L. Improving effect of ligustilide on dextran sodium sulfate-induced ulcerative colitis[J]. Science and Technology of Food Industry,2024,45(4):321−327.] MA G L, XIA X P, MA J L. Improving effect of ligustilide on dextran sodium sulfate-induced ulcerative colitis[J]. Science and Technology of Food Industry, 2024, 45(4): 321−327.
[19] 董晶, 王帅珂, 吴苹, 等. 鱼腥草多酚对葡聚糖硫酸钠(DSS)诱导小鼠溃疡性结肠炎的改善作用[J]. 现代食品科技,2021,37(12):7−13. [DONG J, WANG S K, WU P, et al. Effects of Polygonum hydropiper polyphenols on the improvement of dextran sulfate sodium (DSS)-induced colitis in mice[J]. Modern Food Science and Technology,2021,37(12):7−13.] DONG J, WANG S K, WU P, et al. Effects of Polygonum hydropiper polyphenols on the improvement of dextran sulfate sodium (DSS)-induced colitis in mice[J]. Modern Food Science and Technology, 2021, 37(12): 7−13.
[20] 李杰, 张志旭. 表没食子儿茶素没食子酸酯对葡聚糖硫酸钠诱导的小鼠结肠炎的改善作用[J]. 食品工业科技,2023,44(13):390−397. [LI J, ZHANG Z X. Improving effects of epigallocatechin-3-gallate (EGCG) on dextran sulfate sodium (DSS)-induced colitis[J]. Science and Technology of Food Industry,2023,44(13):390−397.] LI J, ZHANG Z X. Improving effects of epigallocatechin-3-gallate (EGCG) on dextran sulfate sodium (DSS)-induced colitis[J]. Science and Technology of Food Industry, 2023, 44(13): 390−397.
[21] 曲巍, 张智, 马建章, 等. 高通量测序研究益生菌对小鼠肠道菌群的影响[J]. 食品科学,2017,38(1):214−219. [QU W, ZHANG Z, MA J Z, et al. Effect of probiotics on gut microbiota in mice evaluated by high-throughput sequencing[J]. Food Science,2017,38(1):214−219.] QU W, ZHANG Z, MA J Z, et al. Effect of probiotics on gut microbiota in mice evaluated by high-throughput sequencing[J]. Food Science, 2017, 38(1): 214−219.
[22] ZHANG X F, GUAN X X, TANG Y J, et al. Clinical effects and gut microbiota changes of using probiotics, prebiotics or synbiotics in inflammatory bowel disease:A systematic review and meta-analysis[J]. European Journal of Nutrition,2021,60(5):2855−2875. doi: 10.1007/s00394-021-02503-5
[23] JAKUBCZYK D, LESZCZYŃSKA K, GÓRSKA S. The effectiveness of probiotics in the treatment of inflammatory bowel disease (IBD)-A critical review[J]. Nutrients,2020,12(7):1973. doi: 10.3390/nu12071973
[24] TAKEI N, KUDA T, HANDA N, et al. Detection and isolation of typical gut indigenous bacteria in ICR mice fed wheat bran and wheat straw fibre[J]. Food Chemistry,2022,4:100071.
[25] XU H M, HUANG H L, LIU Y D, et al. Selection strategy of dextran sulfate sodium-induced acute or chronic colitis mouse models based on gut microbial profile[J]. BMC Microbiology,2021,21(1):279. doi: 10.1186/s12866-021-02342-8
[26] CAO H Y, LIU J X, SHEN P, et al. Protective effect of naringin on DSS-induced ulcerative colitis in mice[J]. Journal of Agricultural and Food Chemistry,2018,66(50):13133−13140. doi: 10.1021/acs.jafc.8b03942
[27] 苏晓兰, 姚梦茜, 陈琳, 等. 清热化湿调枢方对葡聚糖硫酸钠诱导的结肠炎模型小鼠肠黏膜屏障及相关蛋白表达的影响[J]. 中医杂志,2022,63(16):1566−1572. [SU X L, YAO M Q, CHEN L, et al. Effects of Qingre Huashi Tiaoshu formula on intestinal mucosal barrier and expression of related proteins in dextran sodium sulfate-induced colitis model mice[J]. Journal of Traditional Chinese Medicine,2022,63(16):1566−1572.] SU X L, YAO M Q, CHEN L, et al. Effects of Qingre Huashi Tiaoshu formula on intestinal mucosal barrier and expression of related proteins in dextran sodium sulfate-induced colitis model mice[J]. Journal of Traditional Chinese Medicine, 2022, 63(16): 1566−1572.
[28] CHEN S J, LIU X W, LIU J P, et al. Ulcerative colitis as a polymicrobial infection characterized by sustained broken mucus barrier[J]. World Journal of Gastroenterology,2014,20(28):9468−9475. doi: 10.3748/wjg.v20.i28.9468
[29] FENTON C G, TAMAN H, FLORHOLMEN J, et al. Transcriptional signatures that define ulcerative colitis in remission[J]. Inflammatory Bowel Diseases,2021,27(1):94−105. doi: 10.1093/ibd/izaa075
[30] 王梓宇, 张智慧, 吴鹏, 等. 基于肠道菌群和短链脂肪酸代谢探讨甘草制远志降低肠道炎症的作用机制[J]. 中草药,2023,54(14):4556−4563. [WANG Z Y, ZHANG Z H, WU P, et al. Mechanism of licorice-boiled Polygalae Radix on reducing intestinal inflammation based on intestinal flora and short-chain fatty acid metabolism[J]. Chinese Traditional and Herbal Drugs,2023,54(14):4556−4563.] WANG Z Y, ZHANG Z H, WU P, et al. Mechanism of licorice-boiled Polygalae Radix on reducing intestinal inflammation based on intestinal flora and short-chain fatty acid metabolism[J]. Chinese Traditional and Herbal Drugs, 2023, 54(14): 4556−4563.
[31] YUAN X M, CHEN B Q, DUAN Z L, et al. Depression and anxiety in patients with active ulcerative colitis:Crosstalk of gut microbiota, metabolomics and proteomics[J]. Gut Microbes,2021,13(1):1987779. doi: 10.1080/19490976.2021.1987779
[32] 吴苹, 刘晋倩, 董晶, 等. 鱼腥草多糖对DSS诱导小鼠结肠炎的改善作用[J]. 食品工业科技,2021,42(23):362−369. [WU P, LIU J Q, DONG J, et al. Improving effect of houttuynia cordata polysaccharide on dextran sodium sulfate-induced ulcerative colitis[J]. Science and Technology of Food Industry,2021,42(23):362−369.] WU P, LIU J Q, DONG J, et al. Improving effect of houttuynia cordata polysaccharide on dextran sodium sulfate-induced ulcerative colitis[J]. Science and Technology of Food Industry, 2021, 42(23): 362−369.
[33] 秦妮娜, 马志花, 张晨一, 等. 抗性糊精对急性溃疡性结肠炎小鼠的保护作用[J]. 食品与发酵工业, 2023, 49(5):38−45. [QIN N N, MA Z H, ZHANG C Y, et al. Protective effect of resistant dextrin on mice with acute ulcerative colitis[J]. Food and Fermentation Industries, 2021, 42(23):362−369.] QIN N N, MA Z H, ZHANG C Y, et al. Protective effect of resistant dextrin on mice with acute ulcerative colitis[J]. Food and Fermentation Industries, 2021, 42(23): 362−369.
[34] 卢红, 李明, 李龙明. 广藿香植株水提液的化感自毒作用[J]. 北方园艺,2021,16:108−115. [LU H, LI M, LI L M. Allelotoxic effect of aqueous extract solution from pogostemoncablin[J]. Northern Horticulture,2021,16:108−115.] LU H, LI M, LI L M. Allelotoxic effect of aqueous extract solution from pogostemoncablin[J]. Northern Horticulture, 2021, 16: 108−115.
[35] 韩佩恒. 原儿茶酸增强高脂膳食小鼠肠黏膜屏障功能及护肠固体饮料研发[D]. 南昌:南昌大学, 2023. [HAN P H. Study on the enhancement of intestinal mucosal barrier function of high-fat diet mice by protocatechuic acid and development of intestinal-protective solid beverage[D]. Nanchang:Nanchang University, 2023.] HAN P H. Study on the enhancement of intestinal mucosal barrier function of high-fat diet mice by protocatechuic acid and development of intestinal-protective solid beverage[D]. Nanchang: Nanchang University, 2023.
[36] NIU W, CHEN Y, WANG L, et al. The combination of sodium alginate and chlorogenic acid enhances the therapeutic effect on ulcerative colitis by the regulation of inflammation and the intestinal flora[J]. Food & Function,2022,13(20):10710−10723.
[37] WAN F, ZHONG R, WANG M, et al. Caffeic acid supplement alleviates colonic inflammation and oxidative stress potentially through improved gut microbiota community in mice[J]. Frontiers in Microbiology,2021,12:784211. doi: 10.3389/fmicb.2021.784211





 下载:
下载:

 下载:
下载:
