Effect of Two Different Polysaccharides on the Stability of Emulsion Formed by Rapeseed Oil Bodies
-
摘要: 为了提高油菜籽油体乳液在不同环境条件下的稳定性。本文以带负电荷的可溶性大豆多糖(SSPS)和不带电荷的魔芋葡甘聚糖(KGM)为原料形成油菜籽油体乳液。利用激光粒度分析仪和显微镜分别对两种不同多糖的油菜籽油体乳液进行了粒度分析和微观结构观察,考察了两种含不同多糖的油菜籽油体乳液在不同环境应力(pH、离子浓度和热处理)下的稳定性,以不含多糖的油菜籽油体乳液作为对照。实验结果表明:油菜籽油体乳液的粒径随着SSPS和KGM浓度的增加逐渐减小。除了pH2.0,不含多糖的油菜籽油体乳液在不同pH的粒径都显著高于(P<0.05)含KGM和SSPS的油菜籽油体乳液的粒径,此外,除了pH10.0外,含SSPS的油菜籽油体乳液在不同pH的粒径显著低于(P<0.05)含KGM的油菜籽油体乳液的粒径,而且含SSPS的油菜籽油体乳液的粒径受pH的影响较小。不含多糖的油菜籽油体乳液在不同盐离子浓度时的粒径变化不显著(P>0.05),但都显著高于(P<0.05)含KGM和SSPS的油菜籽油体乳液的粒径。微观结构图也显示了含KGM和SSPS的油菜籽油体乳液在不同盐离子浓度下其油滴的分散性要好于不含多糖的油菜籽油体乳液。不含多糖的油菜籽油体乳液的粒径随着温度的升高而逐渐增加,且都显著高于(P<0.05)含KGM和SSPS的油菜籽油体乳液的粒径,而含SSPS的油菜籽油体乳液的粒径受温度的影响不显著(P>0.05)。综合以上结果,KGM和SSPS都能显著提高油菜籽油体乳液的稳定性,且SSPS效果更好。Abstract: In order to improve the stability of emulsions of rapeseed oil bodies under different environmental conditions. The soluble soybean polysaccharide (SSPS) with negative electric charge and konjac glucomannan (KGM) without electric charge were used to form the emulsions of rapeseed oil bodies. Laser scattering particle analyzer and microscope were used to analyze the particle size and observe the microstructure of rapeseed oil body emulsions. The stability of emulsions of rapeseed oil bodies with two different polysaccharides under different environment stresses (pH, salt concentrations and heat treatments) was investigated, and the emulsions of rapeseed oil bodies without polysaccharide were used as control. The results of experiment showed that the particle of emulsions of rapeseed oil bodies decreased along with the increase of SSPS and KGM concentrations. In addition to pH2.0, the particle size of emulsions of rapeseed oil bodies without polysaccharide at different pH was significantly higher (P<0.05) than that of the emulsions of rapeseed oil bodies with SSPS and KGM. In addition to pH10.0, the particle size of emulsions of rapeseed oil bodies with SSPS was significantly lower (P<0.05) than that of the emulsions of rapeseed oil bodies with KGM. Nevertheless, the pH values caused no effect on the particle size of emulsions of rapeseed oil bodies with SSPS. Although there was no significant changes (P>0.05) for the particle size of emulsions of rapeseed oil bodies without polysaccharide at different salt concentrations, the particle size was significantly higher (P<0.05) than the emulsions of rapeseed oil bodies with SSPS and KGM. The microstructure observation also showed that the oil droplets of emulsions of rapeseed oil bodies with SSPS and KGM had better dispersivity than that of emulsions of rapeseed oil bodies without polysaccharide. The particle size of emulsions of rapeseed oil bodies without polysaccharide increased along with the increase of temperatures, which was significantly higher (P<0.05) than that of the emulsions of rapeseed oil bodies with KGM and SSPS, the temperature changes showed there was no significantly effect (P>0.05) on the particle size of emulsions of rapeseed oil bodies with SSPS. Comprehensive above results, KGM and SSPS can significantly improve the stability of the emulsions of rapeseed oil bodies, SSPS shows a better effect.
-
Keywords:
- soluble soybean polysaccharide /
- konjac glucomannan /
- rapeseed oil bodies /
- emulsions /
- stability
-
油菜籽常常被用作蛋白质和植物油的加工原料,是我国种植面积较大的油料作物之一。油菜籽含有大量粒径为0.5~2.5 μm的球形液滴,该液滴被称为油体。和其他植物种籽油体结构一样,油体内部的甘油三酯基质被磷脂与蛋白质组成的生物膜所包裹,该膜结构与乳脂肪球膜及蛋黄膜结构及组成类似[1−3]。
油体膜表面由多种蛋白质组成,主要有油体蛋白、油体固醇蛋白和油体钙蛋白三种[4]。油体蛋白通常有3个结构域,通过离子键吸附在油体的表面,带负电荷的极性氨基酸残基暴露于外侧,油体蛋白整体表现为亲水性和带负电荷性[5−7]。油体蛋白这一特点有助于形成稳定的油体乳液。除油体蛋白外,其他蛋白对油体乳液的稳定性也起到重要作用[8−10]。油菜籽油体特殊的组成和结构,使得其在形成乳液时不需要额外动力[11]。另外,油体膜表面的蛋白和脂质的种类、结构和含量对油体乳液中油滴表面的电荷、油滴粒径大小和稳定性起着决定性的作用[12]。油菜籽油体膜表面的油体蛋白可以形成空间位阻及静电排斥作用进而阻止油体微球发生融合[13−15]。
油菜籽油体形成的乳液在环境应力下(pH、盐离子和高温)表现不稳定,在一定的pH、盐离子浓度和高温条件下,该乳液油滴会发生聚集。为了提高油菜籽油体乳液的稳定性,选用带负电荷的可溶性大豆多糖(Soluble Soybean polysaccharide,SSPS)和不带电荷的魔芋葡甘聚糖 (Konjac glucomannan,KGM)来稳定油菜籽油体乳液。目前,SSPS是利用新型生物化学技术从豆制品加工的副产物豆渣中分离得到[16]。由于SSPS具有形成稳定乳液的一系列物化特性,除了具有良好的分散性、稳定性和乳化性等特性外,还带有大量的负电荷,具有粘度低、耐酸、耐盐和耐热等一系列的优良物化特性[17−18]。因此,SSPS可以被用来作为提高油菜籽油体乳液的稳定剂,能够确保油菜籽油体乳液在环境应力下(pH、盐离子和高温)保持稳定。KGM是一种从魔芋块茎中提取分离出来的中性不带电荷的水溶性多糖,其分子结构是由β-葡萄糖和α-甘露糖通过β-1,4糖苷键连接而成[19−20]。由于KGM的高吸水性能,可在水中吸水溶胀形成网络结构[21−22]。KGM 还可以作为一种食用纤维促进肠胃蠕动,具有改善肠道菌群,降血脂,辅助减肥的功效[23−24]。KGM 虽然不带电荷,但可以显著提高乳液水相的粘度,而且还具有延迟胃排空及增加饱腹感的作用[25−26]。因此,基于 KGM 的诸多特性,包括粒径、表面电荷、 乳脂稳定性、pH稳定性、热稳定性和冻融稳定性,可以用来增加油菜籽油体乳液水相的粘度,从而提高油菜籽油体乳液的稳定性[26−27]。另外,KGM 在水相中不仅能增加溶液的粘度,而且吸水后其分子结构伸展形成密集的网络结构,该网络结构能将粒径较小的油滴包裹在里面,形成空间阻隔,从而阻碍油滴的聚集,进一步改善油菜籽油体乳液的稳定性[28−29]。
本研究是在油菜籽油体形成的乳液表现不稳定的基础上,通过利用两种特性截然不同的多糖:一种是带负电荷的SSPS;另外一种是不带电荷的KGM。分别从影响乳液的油相和水相两个方面来研究其对油菜籽油体乳液的稳定性。考察SSPS和KGM对油菜籽油体乳液在不同pH、盐离子浓度和热处理条件下的稳定性,以油体乳液的粒径分布、粒径大小、微观结构作为对照来评价SSPS和KGM对菜籽油体乳液稳定性的影响,为油菜籽油体乳液在食品工业中的应用及作为载体运载脂溶性生物活性物质提供必要的理论依据。
1. 材料与方法
1.1 材料与仪器
油菜籽 购自江苏省宿迁市沭阳县种业公司,采收时间为2021年5月;魔芋葡苷聚糖(纯度˃95%) 西安百川生物科技有限公司;可溶性大豆多糖(纯度˃95%) 河南千志商贸有限公司;氯化钠、蔗糖、Tris、氢氧化钠 分析纯,西陇科学股份有限公司。
BD-SW1000光学显微镜 北京普瑞赛司仪器有限公司;L3-5K离心机 湖南凯达科学仪器有限公司;PB-10 pH计 德国Sartorius科技有限公司;800S组织捣碎机 青岛圣吉仪器系统有限公司;Secura125-1CN电子分析天平 Sartorius科技有限公司;HWS-28电热恒温水浴锅 上海一恒科学仪器有限公司;T25 easy clean高速剪切机 广州艾卡仪器设备与限公司;3-18K高速冷冻离心机 美国SIGMA公司;MS-2000激光粒度分析仪 英国Malvern 公司。
1.2 实验方法
1.2.1 油菜籽油体的提取
油菜籽油体的提取参照Liu等[30]的方法。将油菜籽浸泡于蒸馏水中,质量比为1:5,置于 4~6 ℃冰箱中过夜,取出已经充分吸水的油菜籽并沥干,然后用含有0.5 mol/L氯化钠和0.4 mol/L蔗糖的pH7.5 Tris-HCl缓冲溶液(1:5 W/V,50 mmol/L Tris-HCl缓冲溶液)将吸水的油菜籽分散,用组织捣碎机匀浆5 min。用 3 层200目滤布过滤匀浆液,除去滤渣,收集滤液,于4 ℃,10000 r/min的条件下离心45 min,收集上层乳膏状物并均匀分散在上述缓冲溶液中,用上述同样的条件离心45 min,收集上层乳膏状物,再重复离心分离3次,将得到的上层乳膏状物(即油菜籽油体)置于4 ℃冰箱中备用。
1.2.2 含SSPS和KGM的油菜籽油体乳液的制备
准确称取5.0 g油菜籽油体,将5.0 g油菜籽油体分散于95.0 g蒸馏水中,使油菜籽油体占整个乳液质量分数的5%,于400 r/min搅拌30 min,然后用高速均质机于12000 r/min剪切5 min,制成油菜籽油体乳液。
向制备好的油菜籽油体乳液中分别加入质量分数为0.25%、0.5%、1.0%、2.0%、3.0%、4.0%的SSPS;再向另一份制备好的油菜籽油体乳液中分别加入质量分数为0.05%、0.1%、0.15%、0.2%、0.25%、0.3%的KGM。两种含不同多糖的油菜籽油体乳液分别用高速均质机于12000 r/min剪切5 min,分别形成含SSPS和KGM的油菜籽油体乳液。
1.2.3 粒径的测定
不同乳液样品利用激光粒度分析仪测定其粒径。将待测样品用与其pH相同的蒸馏水稀释到合适的浓度,混合均匀后加入样品池。测定时,样品的折射率与水的折射率分别为1.47与1.33,用d43表示样品的平均粒径。
1.2.4 微观结构观察
取2 mL的待观察样品滴在洁净干燥的载玻片中央的位置,用盖玻片轻轻挤压,挤压过程中尽量避免气泡产生,然后调整显微镜的焦距找到目标样品,最后在60倍物镜下进行观察,并拍照。
1.2.5 不同环境条件下乳液的稳定性
1.2.5.1 pH 对乳液稳定性的影响
用1 mol/L的HCL或NaOH将两种含不同多糖的油菜籽油体乳液的pH分别调至2.0、4.0、6.0、8.0和10.0,然后利用激光粒度分析仪分析两种含不同多糖的油菜籽油体乳液在不同pH条件下的粒径,同时利用显微镜对两种含不同多糖的油菜籽油体乳液进行微观结构观察,以不含多糖的油菜籽油体乳液作为对照。
1.2.5.2 离子浓度对乳液稳定性的影响
用1 mol/L的NaCl储备溶液将两种含不同多糖的油菜籽油体乳液的离子浓度分别调至100、200、300、400和500 mmol/L,然后利用激光粒度分析仪分析两种含不同多糖的油菜籽油体乳液在不同盐离子浓度条件下的粒径,同时利用显微镜对两种乳液进行微观结构观察。以不含多糖的油菜籽油体乳液作为对照。
1.2.5.3 温度对乳液稳定性的影响
取一定体积含不同多糖的油菜籽油体乳液分别装入不同试管中,然后分别置于25、35、45、55、65、75、85和95 ℃的恒温水浴锅中加热15 min,取样冷却后用激光粒度分析仪对不同温度下的样品进行粒径分析,同时利用显微对两种含不同多糖的油菜籽油体乳液进行微观结构观察。以不含多糖的油菜籽油体乳液作为对照。
1.3 数据处理
所有实验重复操作3次。实验数据的平均值和方差均通过Origin 8.5分析,数据的差异显著性(P<0.05)使用统计分析软件SPSS进行多重比较分析。
2. 结果与分析
2.1 SSPS对油菜籽油体乳液稳定性的影响
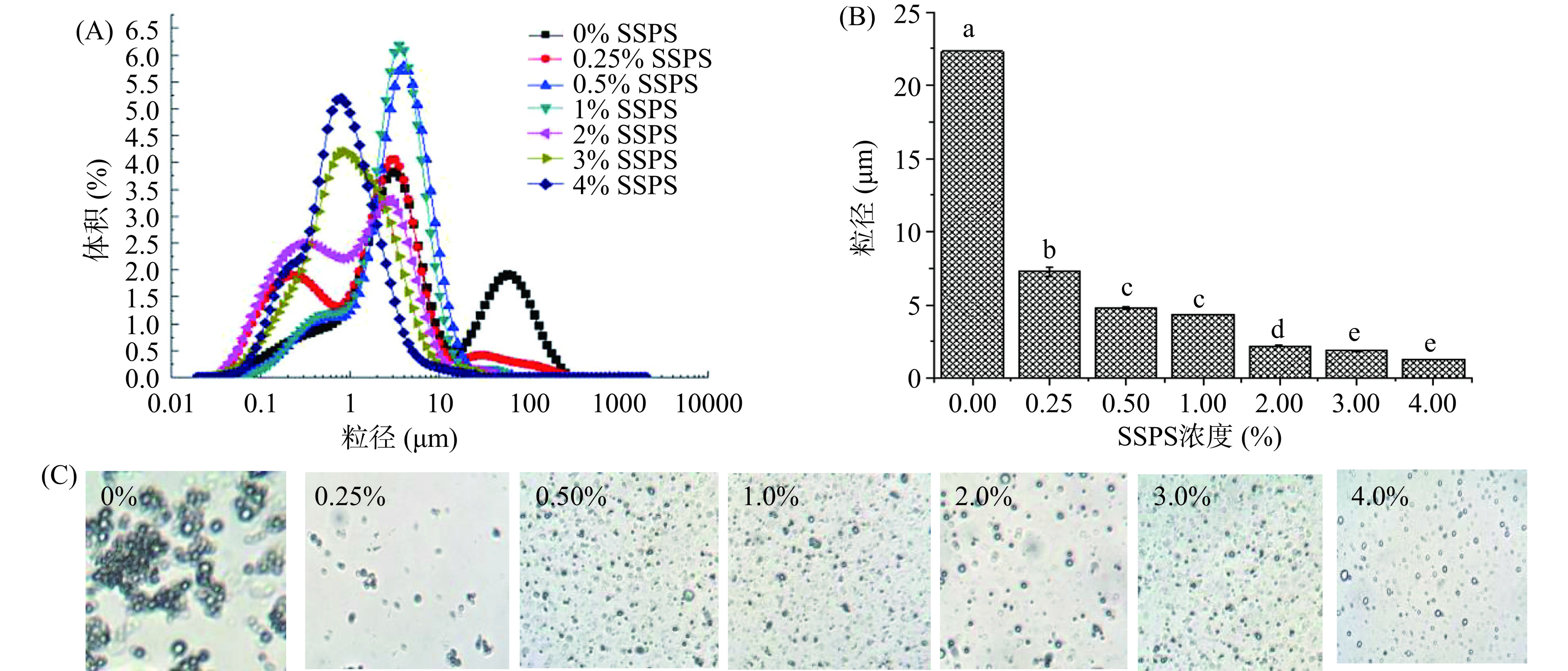
通过向油菜籽油体乳液中添加不同浓度的SSPS (0.0%、0.25%、0.5%、1.0%、2.0%、3.0%、4.0%),以油体乳液的粒径分布、粒径大小和微结构观察来考察SSPS浓度对油菜籽油体乳液稳定性的影响。图1 (A、B和C)是添加不同浓度的SSPS形成的油菜籽油体乳液的粒径分布、粒径大小和微观结构图。从该图可以看出,随着SSPS浓度的增加,油菜籽油体乳液的粒径分布朝着粒径减小的方向变化,说明油菜籽油体乳液的粒径随着SSPS浓度的增加逐渐减小(图1A)。从添加不同浓度的SSPS形成的油菜籽油体乳液的粒径大小图也可以看出,油菜籽油体乳液的粒径随着SSPS浓度的增加,其粒径显著减小(图1B)。图1C也明显地显示了当SSPS浓度较低时(0%和0.25%),油菜籽油体乳液油滴发生了明显的聚集,而随着SSPS浓度的增加,乳液油滴分散性更好,没有聚集现象发生。该结果主要是由于当SSPS浓度较低时,SSPS不足以覆盖于乳液油滴表面,油滴表面电荷较低,静电排斥作用较弱,乳液油滴容易发生聚集,粒径增加,表现出不稳定[30−31]。随着SSPS浓度的增加,油滴表面覆盖的SSPS增加,所带负电荷增加,静电排斥作用增强,油菜籽油体乳液油滴分散性好,不易发生聚集现象,从而保持稳定。添加3.0%和4.0% SSPS的油菜籽油体乳液粒径分别为1.85 μm和1.27 μm,两者之间没有显著性差异(P˃0.05)。根据该实验结果,选取含3.0% SSPS的油菜籽油体乳液作为后期稳定性研究。
2.2 KGM对油菜籽油体乳液稳定性的影响
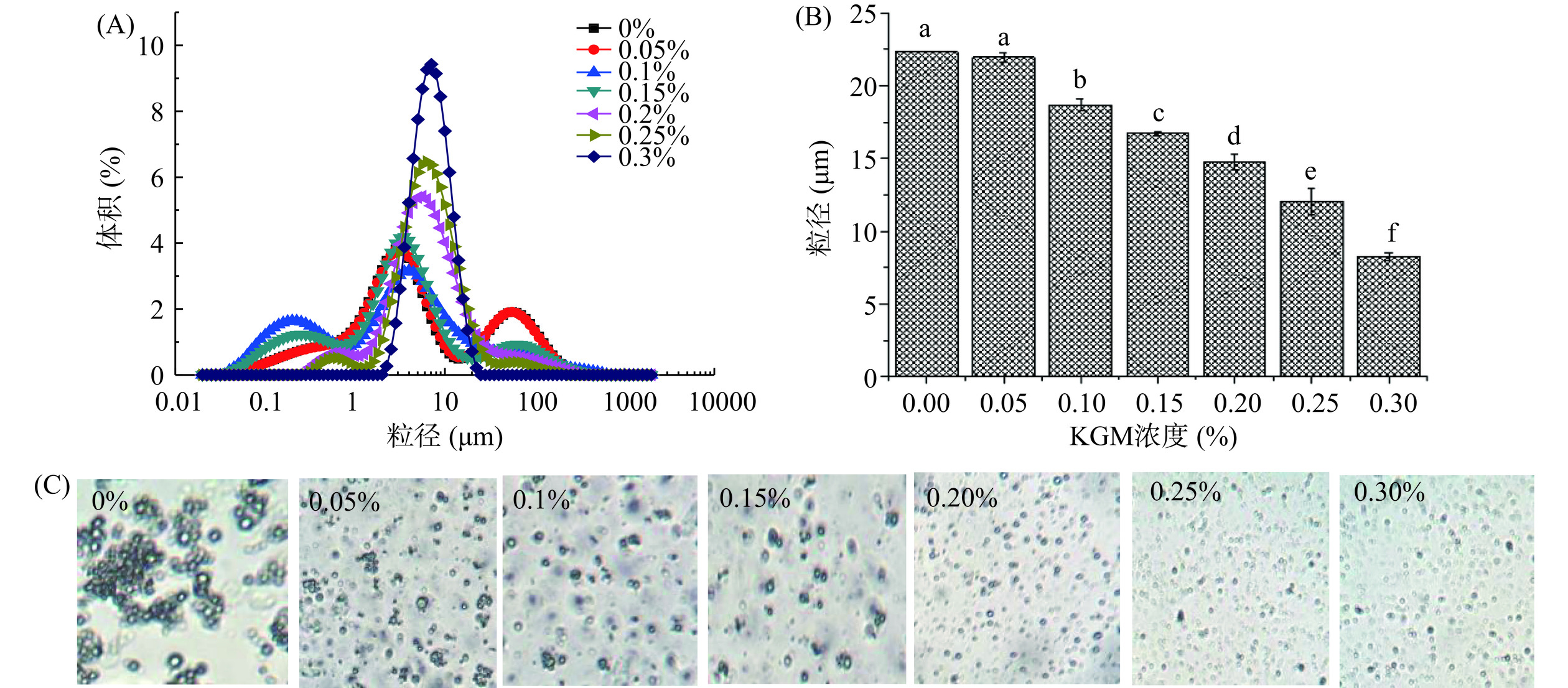
通过向油菜籽油体乳液中添加不同浓度的KGM (0.00%、0.05%、0.10%、0.15%、0.20%、0.25%和0.30%),以油体乳液的粒径分布、粒径大小和微结构来考察KGM对油菜籽油体乳液稳定性的影响。图2 (A、B和C)分别是添加不同浓度的KGM形成的油菜籽油体乳液的粒径分布、粒径大小和微观结构图。从该图可以看出,随着KGM浓度的增加,油菜籽油体乳液的粒径分布朝着粒径减小的方向变化 (图2A)。当KGM添加量为0.30%时,乳液油滴粒径由多峰变为单峰分布,说明其分散更均匀。从添加不同浓度的KGM形成的油菜籽油体乳液的粒径大小图也可以看出,随着KGM浓度的增加油菜籽油体乳液的粒径逐渐减小(图2B)。图2C乳液的微观结构图也显示了当KGM浓度较低时,油菜籽油体乳液的油滴发生了明显的聚集,随着KGM浓度的增加,乳液油滴分散性更好。出现这种结果,主要是随着KGM浓度的增加,乳液水相的黏度增加,降低了乳液油滴的沉降速度,油滴不易聚集,油菜籽油体乳液表现更稳定[32]。根据该实验结果,选取含0.30% KGM的油菜籽油体乳液作为后期稳定性研究。
2.3 两种不同多糖对油菜籽油体乳液稳定性的影响
2.3.1 不同pH对两种不同多糖的油菜籽油体乳液稳定性的影响
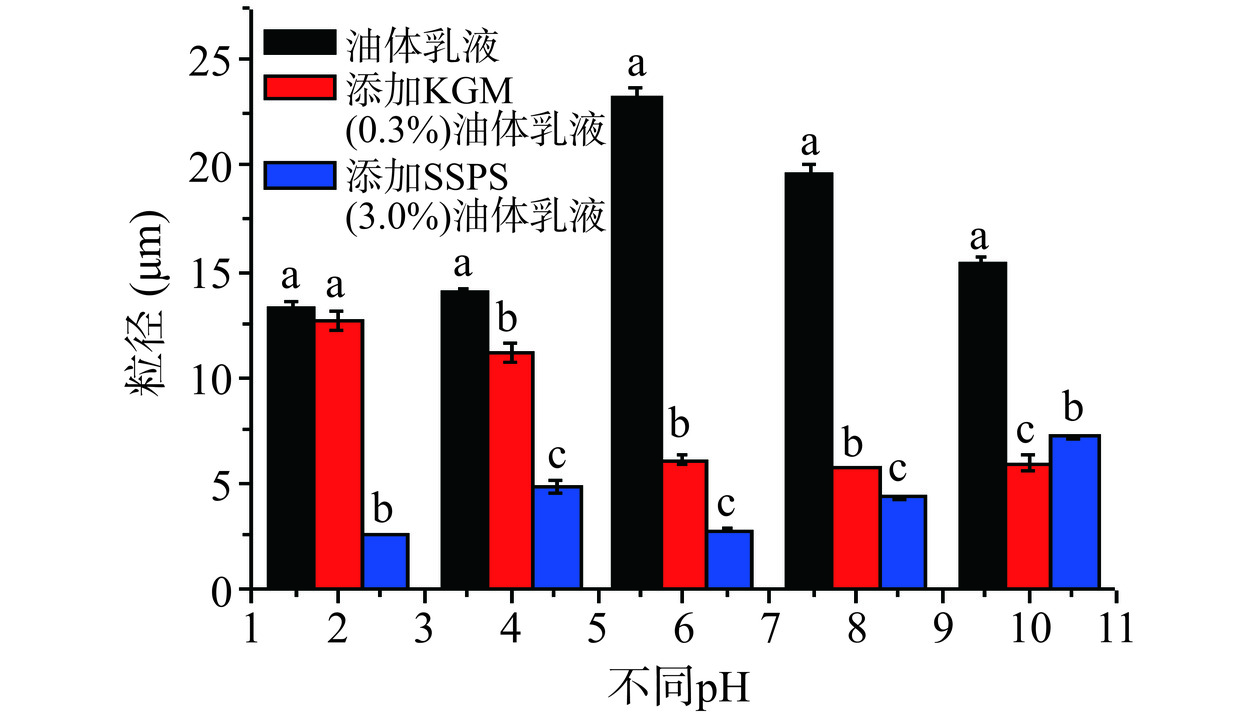
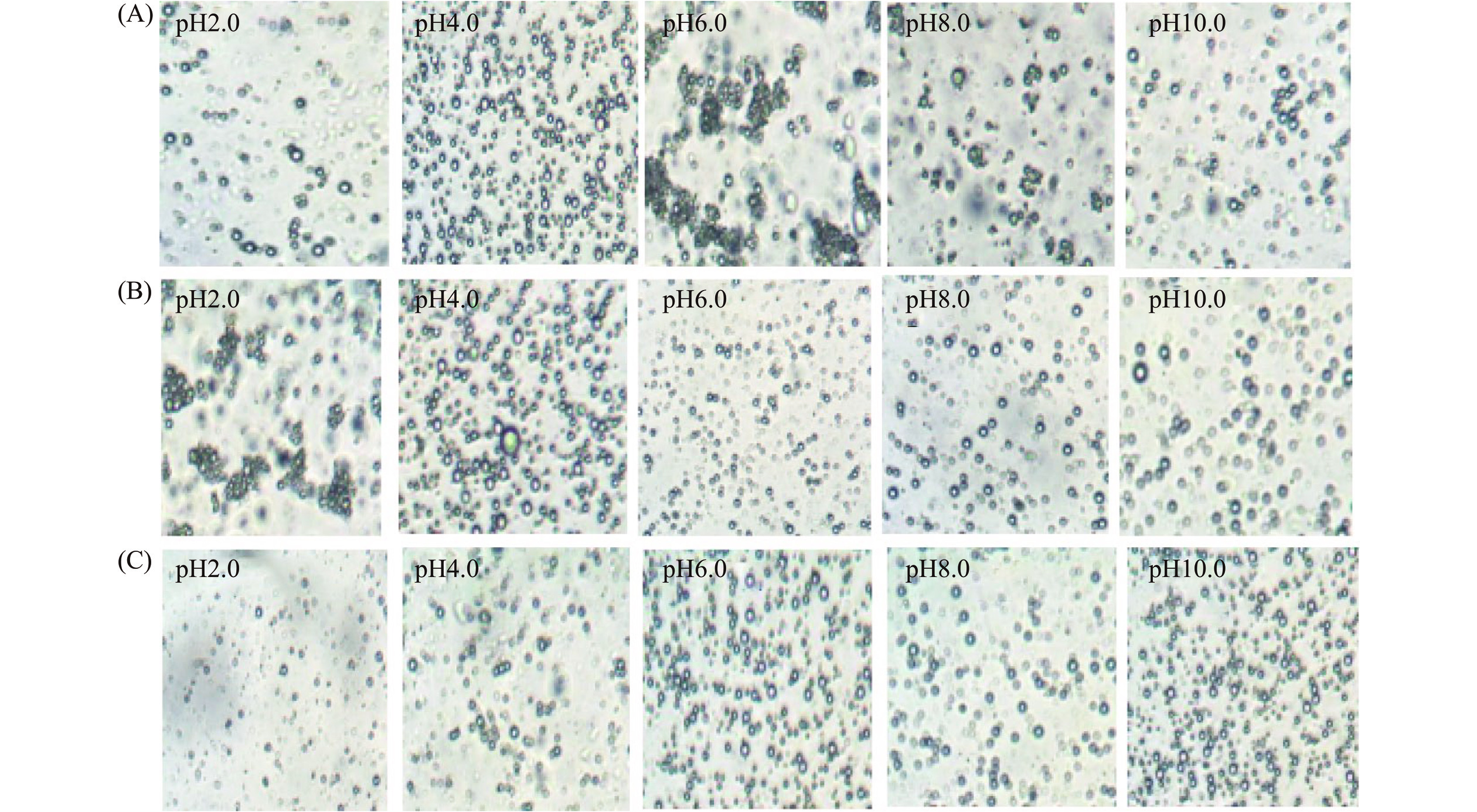
油体乳液在食品加工及胃肠道消化过程中都会受到不同pH的影响,从而影响油体乳液的稳定性和消化特性[19−20]。因此,研究油体乳液在不同pH条件下的稳定性的影响非常必要。图3是含3.0% SSPS的油菜籽油体乳液、含0.30% KGM的油菜籽油体乳液和不含多糖的油菜籽油体乳液在不同pH的粒径大小图。从图3可以看出,不含多糖的油菜籽油体乳液在pH6.0时粒径显著高于(P<0.05)其他pH的粒径,这主要是由于油菜籽油体中蛋白质的等电点pI接近于6.0,此时油菜籽油体乳液油滴表面所带电荷最少,静电排斥作用最弱,油菜籽油体乳液油滴容易聚集,粒径较大,表现出不稳定[33]。另外,除了pH2.0外,不含多糖的油菜籽油体乳液在不同pH的粒径都显著(P<0.05)高于含KGM和SSPS的油菜籽油体乳液的粒径(图3)。图4的微观结构图也显示了不含多糖的油菜籽油体乳液在pH6.0时油滴出现了明显的聚集,表现不稳定。图3显示了含KGM的油菜籽油体乳液的粒径在pH2.0和4.0时有较大的粒径。图4的微观结构图也显示了含KGM的油菜籽油体乳液在pH2.0和4.0时油滴较其他pH聚集更明显。此外,除了pH10.0外,图3还显示了含SSPS的油菜籽油体乳液在不同pH的粒径显著低于(P<0.05)含KGM的油菜籽油体乳液的粒径,而且含SSPS的油菜籽油体乳液的粒径受pH的影响较小。图4的微观结构图也显示了含SSPS的油菜籽油体乳液的油滴在不同pH分散性均较好,没有出现明显的聚集。这种结果可能是SSPS带有大量的负电荷,加之油菜籽油体中本身蛋白所带的负电荷,有足够的负电荷来中和不同pH时的[H+],使乳液在不同pH时仍带有大量的负电荷,乳液油滴之间有较强的静电排斥作用,避免了乳液中油滴的聚集,促进了乳液的稳定性[34]。从以上结果可以看出,KGM和SSPS都能显著提高油菜籽油体不同pH的稳定性,尤其是SSPS效果更好。
2.3.2 不同盐离子浓度对两种不同多糖的油菜籽油体乳液稳定性的影响
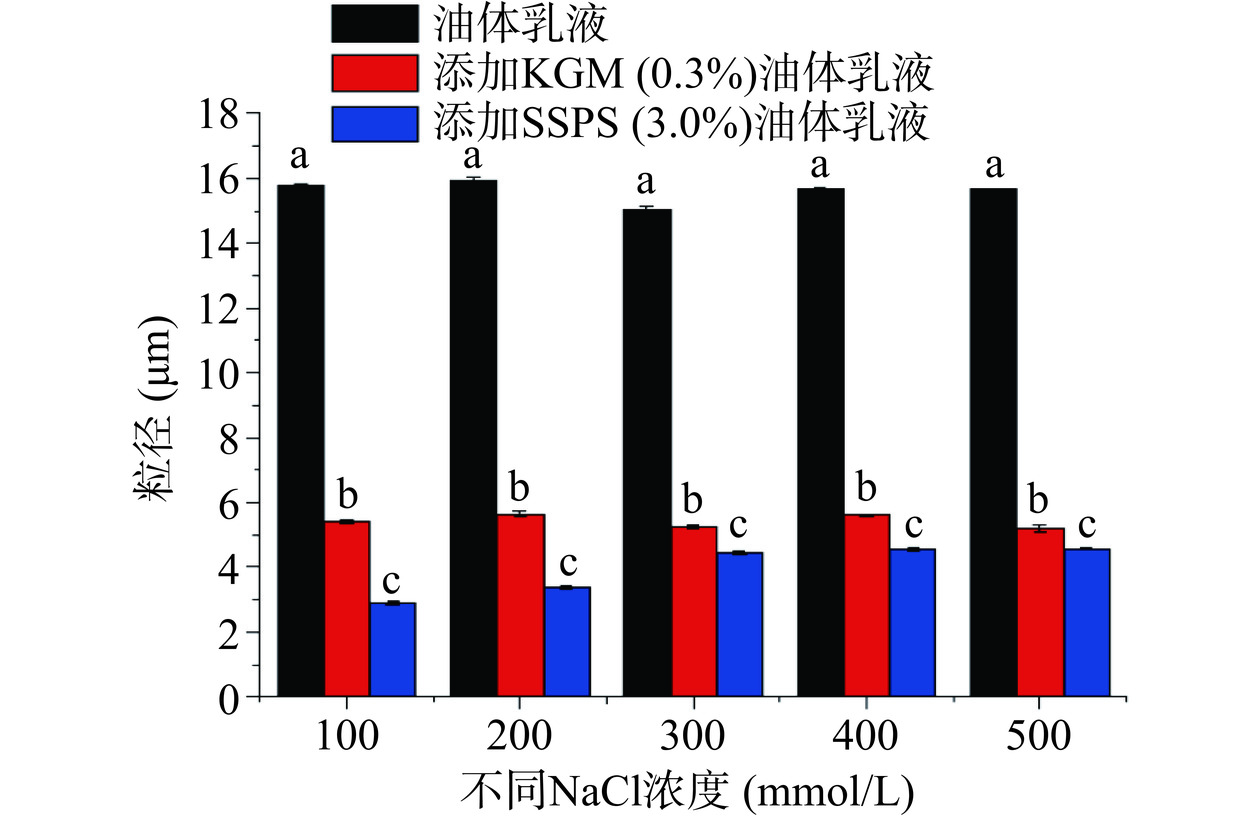
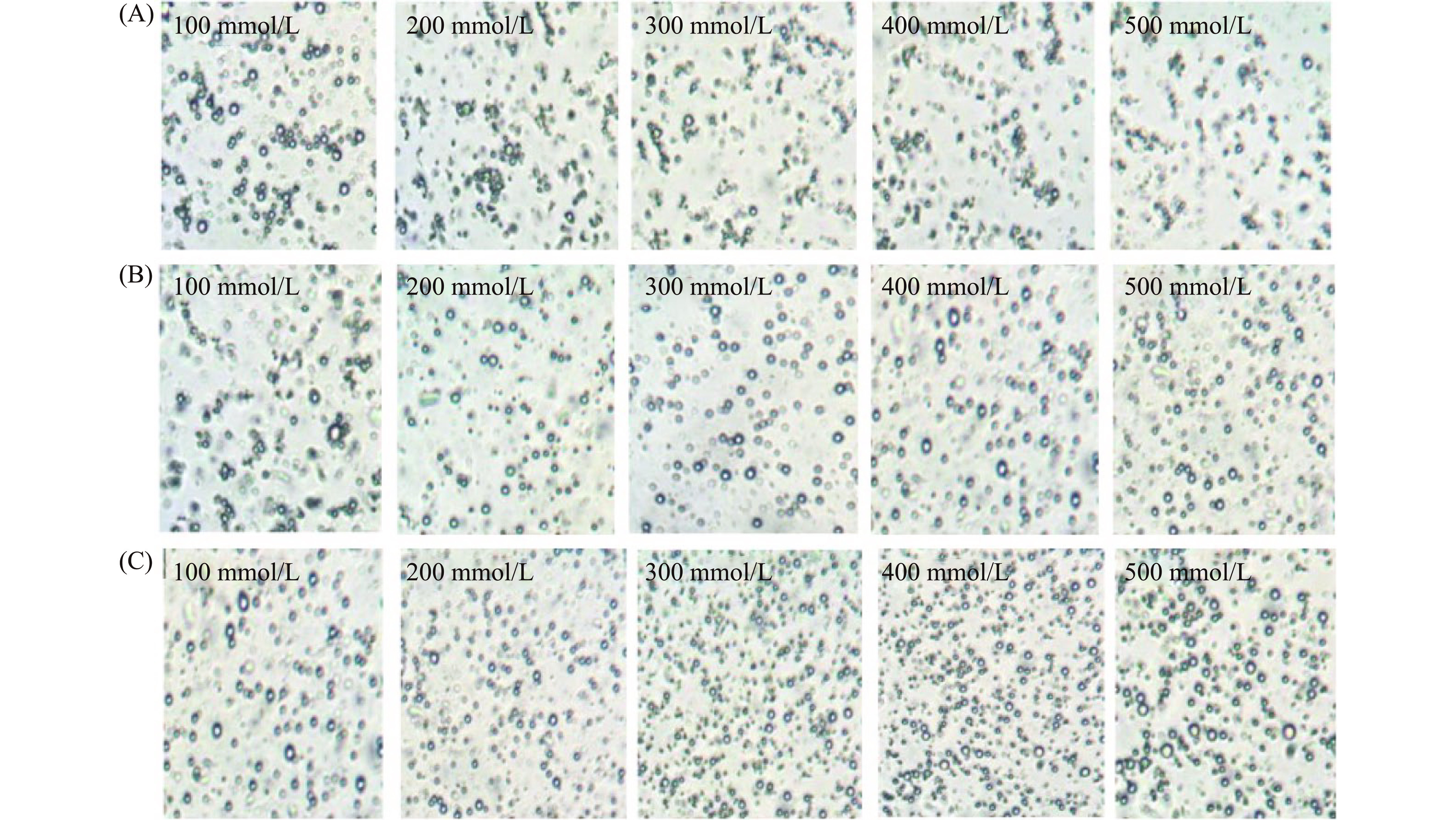
油体乳液在食品加工及胃肠道消化过程中同样会受到盐离子的影响,因此,研究盐离子对两种含不同多糖油菜籽油体乳液稳定性的影响也非常重要[35−36]。图5是两种含不同多糖的油菜籽油体乳液和不含多糖的油菜籽油体乳液在不同盐离子浓度下的粒径大小图。该图显示了不含多糖的油菜籽油体乳液在不同盐离子浓度时的粒径变化不显著(P>0.05),但都显著高于(P<0.05)含KGM和SSPS的油菜籽油体乳液的粒径。图6A的微观结构图也显示了油菜籽油体乳液的油滴受盐离子浓度的影响较小。此外,图5还显示了含KGM的油菜籽油体乳液的粒径受盐离子NaCl浓度的影响较小,图6B的微观结构图也显示了含KGM的油菜籽油体乳液的油滴受盐离子浓度的影响较小。图5还显示了含SSPS的油菜籽油体乳液在不同盐离子浓度时的粒径变化不显著(P>0.05)。图6C微观结构图进一步证实了含SSPS的油菜籽油体乳液在不同盐离子浓度时油滴没有显著性变化。从以上结果可以看出,KGM和SSPS都能显著提高油菜籽油体乳液在不同盐离子浓度下的稳定性。
2.3.3 不同温度对两种不同多糖的油菜籽油体乳液稳定性的影响
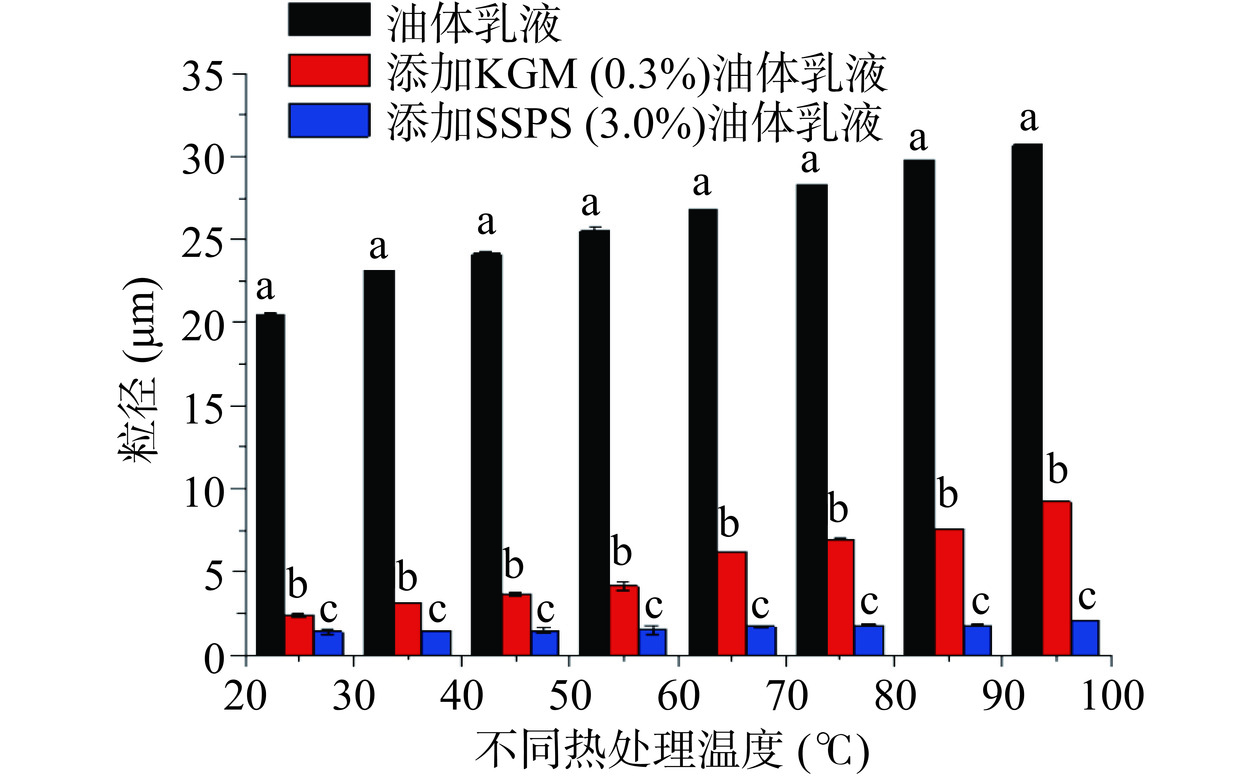
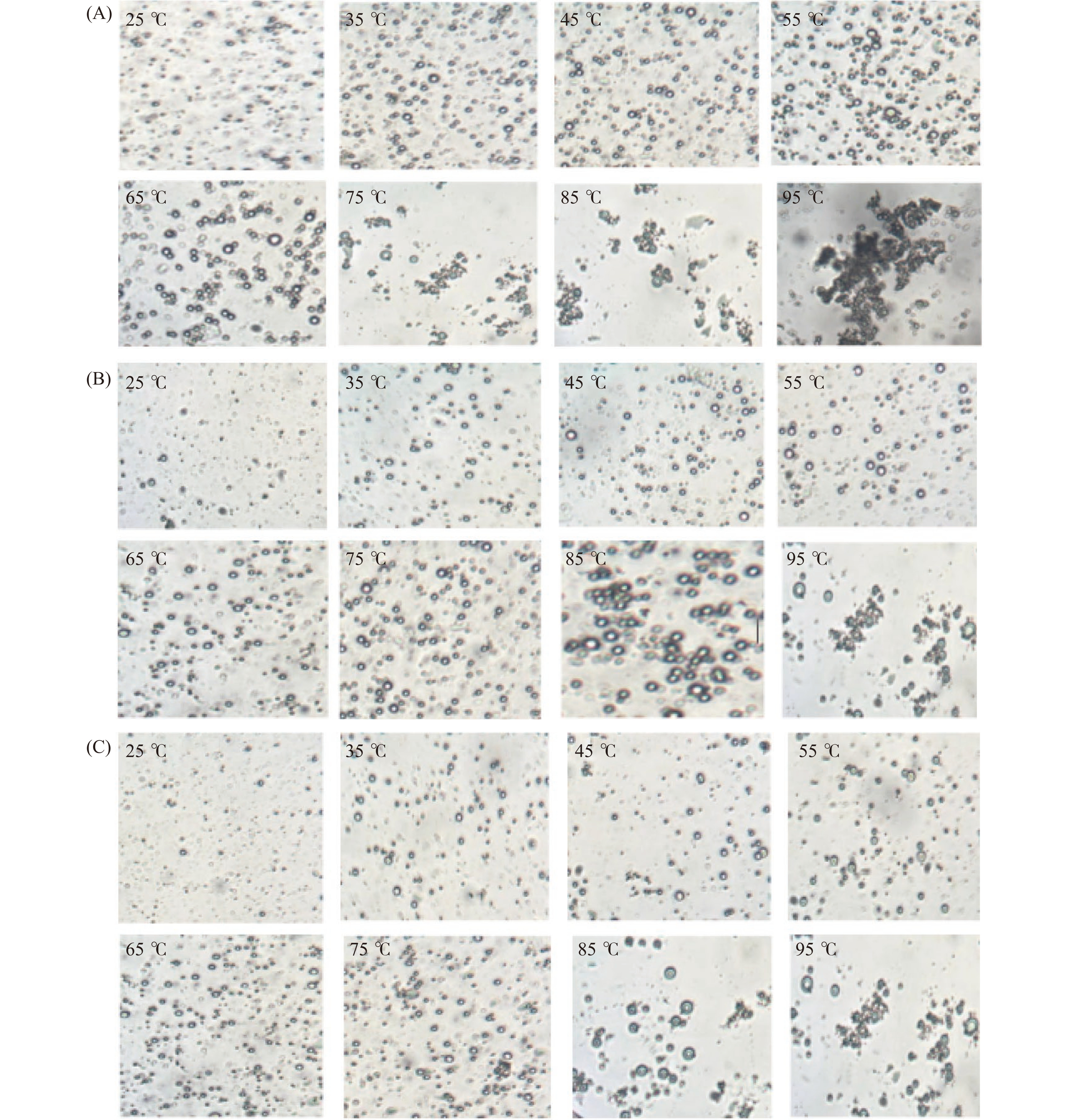
油体乳液能够保持良好的稳定性主要取决于油体中的油体蛋白和油体钙蛋白。油体蛋白和油体钙具有两亲性,对蛋白质的空间结构起到了一定作用,增加了位阻效应,有利于防止油体的聚集[37-38]。加热会导致油体乳液中蛋白质结构的变化,从而影响油体乳液的稳定性。图7 是两种含不同多糖的油菜籽油体乳液和不含多糖的油菜籽油体乳液在不同温度的粒径大小图。该图显示了不含多糖的油菜籽油体乳液的粒径随着温度的升高而逐渐增加,且都显著高于(P<0.05)含KGM和SSPS的油菜籽油体乳液的粒径。图7显示了不含多糖的油菜籽油体乳液在95 ℃时出现了较大粒径的油滴。图8A的微观结构图也显示了不含多糖的油菜籽油体乳液在温度高于75 ℃时出现了油滴明显聚集的现象。该结果可能是由于温度高导致油菜籽油体中蛋白质变性,结构发生了变化,乳化能力下降,乳液表现不稳定[39]。图7也显示了含KGM的油菜籽油体乳液的粒径随着温度的升高,粒径逐渐增大,但显著低于(P<0.05)不含多糖的油菜籽油体乳液的粒径。该结果可能是由于KGM能显著增加油菜籽油体乳液中水相的粘度,阻碍了热传递,从而提高了油菜籽油体乳液的热稳定性[26]。图8B的微观结构图也显示了含KGM的油菜籽油体乳液在温度低于85 ℃表现较稳定,没有油滴聚集现象的发生,而当温度高于85 ℃时乳液油滴出现了聚集的现象。图7同样显示了含SSPS的油菜籽油体乳液在不同温度条件下粒径变化不显著(P>0.05),而且显著低于(P<0.05)不含多糖的油菜籽油体乳液和含KGM的油菜籽油体乳液的粒径。图8C的微观结构图进一步证实了该结果。从以上结果可以看出,KGM和SSPS都能显著提高油菜籽油体乳液的热稳定性,尤其是SSPS的效果更显著。
3. 结论
当油菜籽油体乳液中添加不同浓度的SSPS和KGM时,油菜籽油体乳液的粒径随着SSPS和KGM浓度的增加逐渐减小。且含KGM和SSPS的油菜籽油体乳液的粒径显著低于不含多糖的油菜籽油体乳液的粒径。另外,除了pH10.0外,含SSPS的油菜籽油体乳液在不同pH的粒径显著低于(P<0.05)不含多糖的油菜籽油体乳液和含KGM的油菜籽油体乳液的粒径。含KGM和SSPS的油菜籽油体乳液在不同盐离子浓度时的粒径显著低于(P<0.05)不含多糖的油菜籽油体乳液的粒径。不含多糖的油菜籽油体乳液的粒径随着温度的升高而逐渐增加,且都显著高于(P<0.05)含KGM和SSPS的油菜籽油体乳液的粒径。因此,KGM和SSPS都能显著提高油菜籽油体乳液的稳定性,且SSPS效果更好。该研究对油菜籽油体未来在食品工业中的应用提供了理论基础。然而,两种不同性质的多糖在提高油菜籽油体稳定性的机理方面还需要进一步研究。
-
-
[1] JASON T C T, ANTHONY H C H. Surface structure and properties of plant seed oil bodies[J]. The Journal of Cell Biology,1992,117(2):327−335. doi: 10.1083/jcb.117.2.327
[2] HOU J, FENG X, JIANG M. et al. Effect of NaCl on oxidative stability and protein properties of oil bodies from different oil crops[J]. LWT - Food Science and Technology,2019,113:108263. doi: 10.1016/j.lwt.2019.108263
[3] BOURGEOIS C, GOMAA, A I, LEFEVRE T, et al. Interaction of oil bodies proteins with phospholipid bilayers:A molecular level elucidation as revealed by infrared spectroscopy[J]. International Journal of Biological Macromolecules,2019,122:873−881. doi: 10.1016/j.ijbiomac.2018.10.211
[4] ZAABOUL F, ZHAO Q L, XU Y J. Soybean oil bodies:A review on composition, properties, food applications, and future research aspects[J]. Food Hydrocolloids,2022,124:107296. doi: 10.1016/j.foodhyd.2021.107296
[5] CHIRICO D S, BARI D V, GUZMAN M J R, et al. Assessment of rapeseed oil body (oleosome) lipolytic activity as an effective predictor of emulsion purity and stability[J]. Food Chemistry,2020,316:126355. doi: 10.1016/j.foodchem.2020.126355
[6] JEFF C F C, CORINNE C Y T, TZEN J T C. Cloning and secondary structure analysis of caleosin, a unique calcium-binding protein in oil bodies of plant seeds[J]. Plant and Cell Physiology (Japan),1999,40(10):1079−1086. doi: 10.1093/oxfordjournals.pcp.a029490
[7] ISHII T, MATSUMIYA K, MATSUMURA Y. Combinational effects of acid and salt addition on colloidal, interfacial, and emulsifying properties of purified soybean oil bodies[J]. Food Hydrocolloids,2021,111:106213. doi: 10.1016/j.foodhyd.2020.106213
[8] TAKASHI L S, IKUKO H. Oil-body-membrane proteins and their physiological functions in plants[J]. Biological & Pharmaceutical Bulletin,2010(3):360−363.
[9] PAN Y J, JIN W P, HUANG Q R. Structure, assembly and application of novel peanut oil body protein extracts nanoparticles[J]. Food Chemistry,2022,15(367):130678. doi: 10.1016/j.foodchem.2021.130678
[10] YANG N, FENG Y N, SU C X. Structure and tribology of κ-carrageenan gels filled with natural oil bodies[J]. Food Hydrocolloids,2020,107:105945. doi: 10.1016/j.foodhyd.2020.105945
[11] DAVE A C, YE A Q, SING H. Structural and interfacial characteristics of oil bodies in coconuts (Cocos nucifera L.)[J]. Food Chemistry,2019,15(276):129−139.
[12] 赵路苹. 大豆油体富集物的蛋白质组成及其对油体乳液性质的影响研究[D]. 无锡:江南大学, 2017. [ZHAO L P. Studies on protein composition of soybean oil body concentrate and its effect on properties of oil body emulsion[D]. Wuxi:Jiangnan University, 2017.] ZHAO L P. Studies on protein composition of soybean oil body concentrate and its effect on properties of oil body emulsion[D]. Wuxi: Jiangnan University, 2017.
[13] IDOGAWA S, ABE N, ABE K, et al. Effect of oleosins on the stability of oil bodies in soymilk[J]. Food Science and Technology Research,2018,24(4):677−685. doi: 10.3136/fstr.24.677
[14] NIKIFORIDIS C V, SCHOLTEN E. High internal phase emulsion gels (HIPE-gels) created through assembly of natural oil bodies[J]. Food Hydrocolloids,2015,43:283−289. doi: 10.1016/j.foodhyd.2014.05.030
[15] TZEN J, CAO Y, LAURENT P, et al. Lipids, proteins, and structure of seed oil bodies from diverse species[J]. Plant Physiology,1993,101:267−276. doi: 10.1104/pp.101.1.267
[16] 孙敏, 陈玮, 高红亮, 等. 大豆水溶性多糖的提取及其对酸性乳饮料的稳定作用[J]. 西北农林科技大学学报(自然科学版),2007,35(1):106−109. [SUN M, CHEN W, GAO H L, et al. Extraction of water soluble soybean polysaccharides and its application in acid dairy beverages[J]. Journal of Northwest A δ F university (Nat Sci Ed),2007,35(1):106−109.] doi: 10.3321/j.issn:1671-9387.2007.01.023 SUN M, CHEN W, GAO H L, et al. Extraction of water soluble soybean polysaccharides and its application in acid dairy beverages[J]. Journal of Northwest A δ F university (Nat Sci Ed), 2007, 35(1): 106−109. doi: 10.3321/j.issn:1671-9387.2007.01.023
[17] 李庄. 大豆水溶性多糖的提取与应用研究[D]. 上海:华东师范大学, 2005. [LI Z. Extraction and application research of soluble soybean polysaccharides[D]. Shanghai:East China Normal University, 2005.] LI Z. Extraction and application research of soluble soybean polysaccharides[D]. Shanghai: East China Normal University, 2005.
[18] 张倩. 可溶性大豆多糖的提取、鉴定及生物活性的研究[D]. 合肥:合肥工业大学, 2007. [ZHANG Q. Extraction, appraised and immunological activity assay of soluble soybean polysaccharides[D]. Hefei:Hefei University of Technology, 2007.] ZHANG Q. Extraction, appraised and immunological activity assay of soluble soybean polysaccharides[D]. Hefei: Hefei University of Technology, 2007.
[19] JIN W P, GE H H, WANG Y T, et al. Molecular migration of konjac glucomannan and gum Arabic phase separation and its application in oil-water interfaces[J]. Food Hydrocolloids,2016,61:868−876. doi: 10.1016/j.foodhyd.2016.07.010
[20] CHUA M, BALDWIN T C, HOCKING T J, et al. Traditional uses and potential health benefits of amorphophallus konjac K. Koch ex N. E. Br[J]. Journal of Ethnopharmacology,2010,128(2):268−278. doi: 10.1016/j.jep.2010.01.021
[21] LIN W M, NI Y S, WANG L, et al. Physicochemical properties of degraded konjac glucomannan prepared by laser assisted with hydrogen peroxide[J]. International Journal of Biological Macromolecules,2019,129(15):78−83.
[22] ZHANG Y, ZHONG Q X. Multiple-layered coatings on L-glutamine solid microparticles for the retention during storage and enteric delivery during in vitro digestions[J]. Food Hydrocolloids,2015,43:584−592.
[23] ARVILL A, BODIN L. Effect of short-term ingestion of konjac glucomannan on serum cholesterol in healthy men[J]. American Journal of Clinical Nutrition,1995,61(3):585−589. doi: 10.1093/ajcn/61.3.585
[24] ZHANG Y. Study on purification technology of glucomannan from amorphophallus konjac and its antioxidation[J]. Food Research and Development,2015,36(4):87−91.
[25] SHANG L C, WANG Y, REN Y Y, et al. In vitro gastric emptying characteristics of konjac glucomannan with different viscosity and its effects on appetite regulation[J]. Food & Function,2020,11(9):7596−7610.
[26] LU W, ZHENG B D, MIAO S. Improved emulsion stability and modified nutrient release by structuring O/W emulsions using konjac glucomannan[J]. Food Hydrocolloids,2018,81:120−128. doi: 10.1016/j.foodhyd.2018.02.034
[27] 王雅立. 魔芋葡甘聚糖分子稳定性研究[D]. 福州:福建农林大学, 2007. [WANG Y Y. Stable research of konjac glucomannan molecular[D]. Fuzhou:Fujian Agriculture and Forestry University, 2007.] WANG Y Y. Stable research of konjac glucomannan molecular[D]. Fuzhou: Fujian Agriculture and Forestry University, 2007.
[28] HE Y, WANG S S, LI J, et al. Interaction between konjac glucomannan and tannic acid:Effect of molecular weight, pH and temperature[J]. Food Hydrocolloids,2019,94:451−458. doi: 10.1016/j.foodhyd.2019.03.044
[29] LI J, LI B, GENG P, et al. Ultrasonic degradation kinetics and rheological profiles of a food polysaccharide (konjac glucomannan) in water[J]. Food Hydrocolloids,2017,70:14−19. doi: 10.1016/j.foodhyd.2017.03.022
[30] LIU C H, WANG R C, HE S H. et al. The stability and gastro-intestinal digestion of curcumin emulsion stabilized with soybean oil bodies[J]. LWT-Food Science and Technology,2020,131:109963. doi: 10.1016/j.lwt.2020.109663
[31] WU N N, YANG X Q, TENG Z, et al. Stabilization of soybean oil body emulsions using κ, ι, λ-carrageenan at different pH values[J]. Food Research International,2011,44(4):1059−1068. doi: 10.1016/j.foodres.2011.03.019
[32] MCCLEMENTS D J. Food emulsions:Principles, practice, and techniques[M]. CRC Press:Boca Raton, 2005.
[33] HE S H, GU C, WANG D G, et al. The stability and in vitro digestion of curcumin emulsions containing konjac glucomannan[J]. LWT-Food Science and Technology,2020,117:108672. doi: 10.1016/j.lwt.2019.108672
[34] 何胜华, 邓乾春. 大豆和油菜籽油体形成的乳液的稳定性及胃肠道消化特性[J]. 食品科学,2021,13(42):34−42. [HE S H, DENG Q C. Stability and in vitro simulated gastrointestinal digestion properties of natural soybean and rapeseed oil body emulsions[J]. Food Science,2021,13(42):34−42.] HE S H, DENG Q C. Stability and in vitro simulated gastrointestinal digestion properties of natural soybean and rapeseed oil body emulsions[J]. Food Science, 2021, 13(42): 34−42.
[35] CÉSAR B D, TRANDY W, ANA M M. Multilayer emulsions stabilized by vegetable proteins and polysaccharides[J]. Current Opinion in Colloid & Interface Science,2016,25:51−57.
[36] 龚凌霄, 曹文燕, 王静, 等. 动态智能人体消化模拟体系及其在食品研究领域中的应用[J]. 中国食品学报,2018,18(10):258−268. [GONG L X, CAO W Y, WANG J. et al. Advanced in dynamic multicompartmental gastrointestinal tract models and its food application[J]. Journal of Chinese Institute of Food Science and Technology,2018,18(10):258−268.] doi: 10.16429/j.1009-7848.2018.10.034 GONG L X, CAO W Y, WANG J. et al. Advanced in dynamic multicompartmental gastrointestinal tract models and its food application[J]. Journal of Chinese Institute of Food Science and Technology, 2018, 18(10): 258−268. doi: 10.16429/j.1009-7848.2018.10.034
[37] AOKI T, DECKER E A, MCCLEMENTS D J. Influence of environmental stresses on stability of O/W emulsions containing droplets stabilized by multilayered membranes produced by a layer-by-layer electrostatic deposition technique[J]. Food Hydrocolloids,2005,19:209−220. doi: 10.1016/j.foodhyd.2004.05.006
[38] CHEN Y, ONO T. Simple extraction method of non-allergenic intact soybean oil bodies that are thermally stable in an aqueous medium[J]. Journal of Agricultural and Food Chemistry,2010,58:7402−7407. doi: 10.1021/jf1006159
[39] ZHANG S, CHEN H J, GENG F, et al. Natural oil bodies from typical oilseeds structural characterization and their potentials as natural delivery system for curcumin[J]. Food Hydrocolloids,2022,128:107521. doi: 10.1016/j.foodhyd.2022.107521





 下载:
下载:
 下载:
下载:
